1

ACC | 1-Aminocyclopropane-1-carboxylic acid
AS11 1800 | Clonality: Polyclonal | Host: Rabbit | Reactivity: ACC
- Product Info
-
Immunogen: KLH-conjugated ACC
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 200 �l Reconstitution: For reconstitution add 200 �l of sterile water Storage: Store lyophilized/reconstituted at -20�C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Immunogold (IG) Recommended dilution: 1 : 100 (IG) - Reactivity
-
Confirmed reactivity: ACC | 1-Aminocyclopropane-1-carboxylic acid Predicted reactivity: ACC | 1-Aminocyclopropane-1-carboxylic acid Not reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
application example
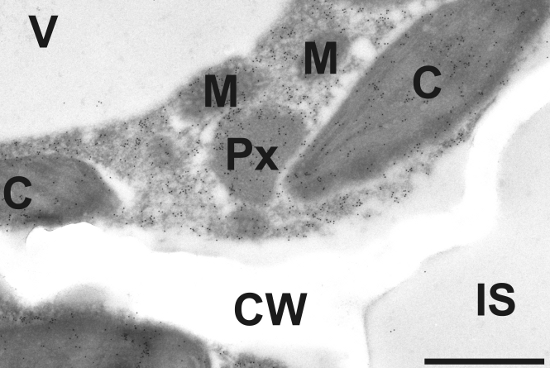
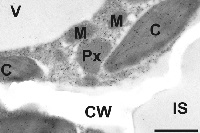
Image shows gold particle density in Arabidopsis thaliana Col-0 after immunogold labeling of ACC in the transmission electron microscope. Gold particles could be detected in chloroplasts (C), mitochondria (M), peroxisomes (Px), and the dense cytosol. No or very few gold particles bound to ACC could be detected in vacuoles (V), intercellular spaces (IS) and cell walls (CW). Bar=1µm.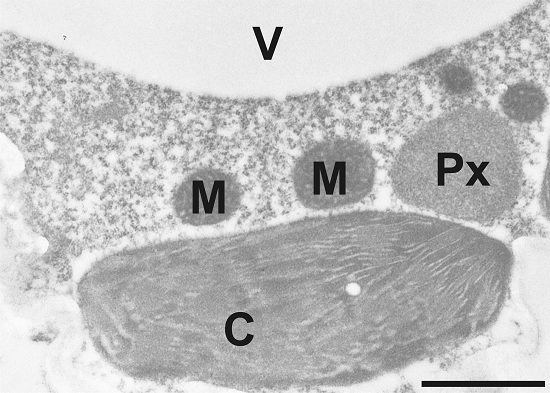
Image shows leaf cell of Arabidopsis thaliana Col-0 treated with preimmune serum instead of the primary antibody against ACC in the transmission electron microscope. No or only very few gold particles were present on the sections. (C) chloroplast, (M) mitochondria, (Px) peroxisome. Bar=1µm.
Sample fixation: Samples for cytohistochemical analysis were fixed for 90 minutes in 2.5% paraformaldehyde/0.5% glutardialdehyde in 0.06M Sørensen phosphate buffer (pH 7.2). Samples were rinsed 4 times for 10 minutes in phosphate buffer then dehydrated with increasing concentrations of acetone (50%, 70% and 90% 20 min for each step). The samples were then transferred to increasing concentrations of LR-White resin (30%, 60%, 100%; London Resin Company Ltd., Berkshire, UK) mixed with acetone (90%) for at least 3 h at each step at RT. Finally, the samples were embedded in pure, fresh LR-White resin and polymerized at 50°C for 48 h in small plastic cups under anaerobic conditions.
Immunogold labeling for electron microscopy: Block ultrathin sections (80nm) prepared for immunogold labeling with 2% bovine serum albumine (BSA) in phosphate buffered saline (PBS, pH 7.2). Then treat the sections with the primary antibody diluted 1:100 in PBS containing 1% BSA for 2 h at room temperature. After a short rinse in PBS (3 X 5 min), incubate samples with a gold-conjugated secondary antibody (goat anti-rabbit IgG; eg. 10nm; British BioCell International, Cardiff, UK) diluted 1:50 in PBS including 1% BSA for 90 min at room temperature. After a short wash in PBS (2 X 5 min), and distilled water (3 X 5 min) observe grids under a transmission electron microscope.
Courtesy of Dr. Bernd Zechmann, Graz University, Austria
- Additional Information
-
Additional information (application): Details of used immunogold protocol are provided under an image - Background
-
Background: ACC (1-Aminocyclopropane-1-carboxylic acid) plays an important role in the biosynthesis of the plant hormone ethylene. It is synthesized by the enzyme ACC synthase ( EC 4.4.1.14) from methionine and converted to ethylene by ACC oxidase (EC 1.14.17.4).
- Product Citations
-
Selected references: Wilmowicz et al. (2019). Abscisic acid and ethylene in the control of nodule-specific response on drought in yellow lupine. Environmental and Experimental Botany Available online 2 October 2019, 103900.
Serova et al. (2018). Early nodule senescence is activated in symbiotic mutants of pea (Pisum sativum L.) forming ineffective nodules blocked at different nodule developmental stages. Protoplasma. 2018 Apr 3. doi: 10.1007/s00709-018-1246-9. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Agrisera Educational Posters Collection
- Reviews:
-
Agata Ku?ko | 2021-02-05Excellent product to detect ACC in plant tissues. We applied this Ab for ACC localization in roots, as well as abscission zone fragments of yellow lupine flowers. Great results have been observed.Wilmowicz E., Ku?ko A., Goli?ska P., Burchardt S., Przywieczerski T., ?widzi?ski M., Brzozowska P., Kapu?ci?ska D. (2020) Abscisic acid and ethylene in the control of nodule-specific response on drought in yellow lupine. Environ. Exp. Bot. 169: 10390Wilmowicz E.*, Ku?ko A.*, Burchardt S., Przywieczerski T. (2019) Molecular and hormonal aspects of drought-triggered flower shedding in yellow lupine. International Journal of Molecular Sciences, 20: 3731.Ku?ko A., Wilmowicz E., Pokora W., Alch� J. (2020) Disruption of auxin gradient in abscission zone area evokes asymmetrical changes leading to flower separation in yellow lupine. International Journal of Molecular Sciences, 21: 3815.Ku?ko A., Wilmowicz E., Ostrowski M. (2019) Spatio-temporal IAA gradient is determined by interactions with ET and governs flower abscission. Journal of Plant Physiology 236: 51-60. Frankowski K., Ku?ko A., Zienkiewicz A., Zienkiewicz K., de Dios Alch� J., Kopcewicz J., Wilmowicz E. (2017) Ethylene-dependent effects on generative organ abscission of Lupinus luteus. Acta Soc. Bot. Pol. 86: 3540

