1
NPR1 | Nonexpresser of PR genes 1
AS12 1854 Clonality: Polyclonal Host: Rabbit | Reactivity: Arabidopsis thaliana
- Data sheet
- Product Info
-
Immunogen: KLH-conjugated peptide, chosen from NPR1 sequence of Arabidopsis thaliana, TAIR: AT1G64280, UniProt: P93002 Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles,Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 1000 (WB) Expected | apparent MW: 66 | 66 kDa
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana Predicted reactivity: Arabidopsis thaliana Not reactive in: Marchantia sp. ,Nicotiana benthamiana, Nicotiana tabacum, Solanum lycopersicum, Solanum tuberosum
- Application Examples
-
application example
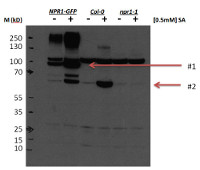
Arabidopsis thaliana samples (0.2 g) were collected from leaf tissue (3 week old rosettes; 24 hours after +/- 0.5 mM sodium salicylate spray). Total protein was extracted in a buffer containing 50 mM Tris-Hcl, pH 7.5; 150 mM NaC; 0.5 mM EDTA; 0.1 % Triton X-100; 0.2 % nonidet P-40; 50 uM MG115 and samples were adjusted to equal total protein concentration. Samples were denatured with 4X SDS Sample buffer with 200 mM DTT (final sample concentration of 50 mM) at 75 C for 15 min. Protein samples (30 µg of total protein) were separated on 4-12% Bis-Tris gel and blotted to PVDF using an iBlot (semi-dry transfer system; Life technologies). The membrane was blocked with 1X PBS-T containing 5% low fat dry milk ans 0.1 % tween-20, for 1h at room temperature (RT) with agitation and incubated in the primary antibody (in blocking solution) at a dilution of 1: 1 000 over night at 4 C with agitation. The antibody solution was decanted and the membrane was washed with blocking solution, 3 times for 10 min each at RT with agitation. The membrane was then incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated) diluted to 1:10 000 in blocking solution for 1h at RT with agitation. The membrane was washed as described above, rinsed with 1x PBS-T and developed for 2 min with Pierce West Dura extended duration substrate. Exposure time was 30 seconds.
** Note: 50 mM DTT concentration in the extraction buffer is necessary to reduce NPR1 oligomer formation.
** Note: The expression level of endogenous NPR1 is very low in extracts from healthy, untreated plants. A successful SA treatment or pathogen infection is absolutely necessary for good detection.
Courtesy Dr. John Withers, Duke University, USAApplication examples: 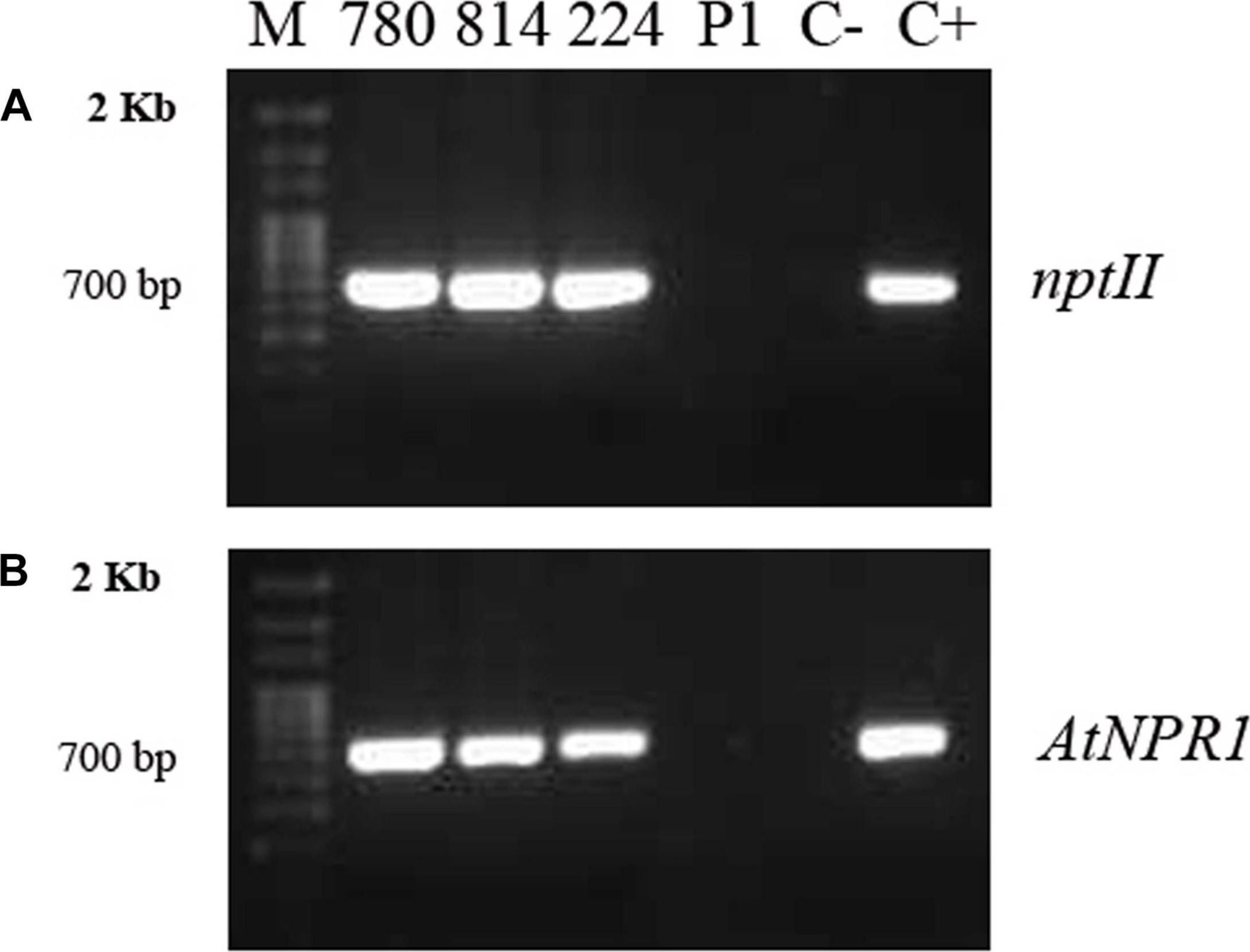
Reactant: Olea europaea (Olive)
Application: Western Blotting
Pudmed ID: 32265961
Journal: Front Plant Sci
Figure Number: 2A,B
Published Date: 2020-04-09
First Author: Narvaez, I., Pliego Prieto, C., et al.
Impact Factor: 5.435
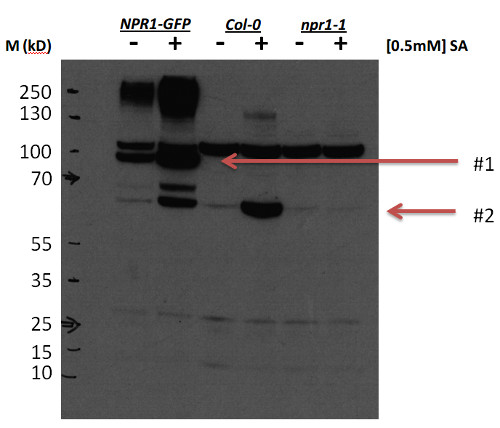
Open PublicationPCR amplifications of the nptII(A) and AtNPR1(B) gene fragments from genomic DNA extracted from embryogenic callus from the different transgenic AtNPR1 lines and non-transformed line P1.780, 814, and 224: transgenic AtNPR1 lines; P1, non-transgenic control; C–, negative control (without DNA); C+, 35S, AtNPR1 binary plasmid; M, molecular marker.
- Additional Information
-
Additional information: For successful detection using NPR1 antibody please follow protocol suggested below. NPR1 protein readily oligomerizes, in addition to any naturally occurring oligomer, during extraction. Therefore 50 mM DTT has to be used as well as denaturation at 75°C for 15 minutes.
Engogenous NPR1 level is very low, thereofre SA treatment is absolutely necessary for good detection.
This antibody is recognizing NPR1-GFP in the 35S overexpression line.Additional information (application): Please note that depending upon detection system you are using, longer exposure time may be required with this antibody, Chemiluminescent detection reagent in extreme femtogram range is necessary - Background
-
Background: NPR1 (regulatory protein NPR1) is a key positive regulator of the SA-dependent signaling pathway that negatively regulates JA-dependent signaling pathway. Controls the onset of systemic acquired resistance (SAR). Subcellular localization: cytoplasm, nucleaus (following induction by salicylic acid treatment or after pathogen infection. Alternative names: BTB/POZ domain-containing protein NPR1, Non-inducible immunity protein 1, Nim1, Salicylic acid insensitive 1, Sai1, ATNPR1.
- Product Citations
-
Selected references: Yao et al. (2023). High air humidity dampens salicylic acid pathway and NPR1 function to promote plant disease.EMBO J. 2023 Nov 2;42(21):e113499.doi: 10.15252/embj.2023113499.
Arenas-Alfonseca et al. (2021) Arabidopsis beta-cyanoalanine synthase mutation overcomes NADPH oxidase action in response to pathogens. J Exp Bot. 2021 Mar 26:erab137. doi: 10.1093/jxb/erab137. Epub ahead of print. PMID: 33770168.
Nomoto et al. (2021) Suppression of MYC transcription activators by the immune cofactor NPR1 fine-tunes plant immune responses. Cell Rep. 2021 Dec 14;37(11):110125. doi: 10.1016/j.celrep.2021.110125. PMID: 34910911.
Lei et al. (2020). Construction of gold-siRNANPR1 nanoparticles for effective and quick silencing of NPR1 in Arabidopsis thaliana. DOI: 10.1039/D0RA02156C (Paper) RSC Adv., 2020, 10, 19300-19308 - Reviews:
-
This product doesn't have any reviews.
Accessories
AS10 687 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, H. vulgare, N. bentamiana, S. oleracea, S. lycopersicum, T. aestivum, Z. mays, V. vinifera


