1
Lhcb1-P | LHCII type I chlorophyll a/b-binding protein, phopshorylated
AS13 2704 | Clonality: Polyclonal | Host: Rabbit | Reactivity:phosphorylated Lhcb1 from Arabidopsis thaliana, Echinochloa crus-galli,, Oryza sativa, Panicum miliaceum, Pisum sativum, Zea mays
- Product Info
-
Immunogen: KLH-conjugated synthetic peptide RKT*VAKPKGP, where T* indicates phospho-Thr
Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 25 µg Reconstitution: For reconstitution add 25 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 10 000 (WB) Expected | apparent MW: 25 | 25 kDa for Arabidopsis thaliana
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Echinochloa crus-galli,, Oryza sativa, Panicum miliaceum, Pisum sativum, Zea mays
Predicted reactivity: Arachis hypogaea, Colobanthus quitensis Kunt Bartl, Hordeum vulgare, Mesembryanthemum crystallinum, Nicotiana tabacum, Phaseolus vulgaris, Silene vulgaris, Solanum lycopersicum, Spinacia oleracea,
Species of your interest not listed? Contact usNot reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
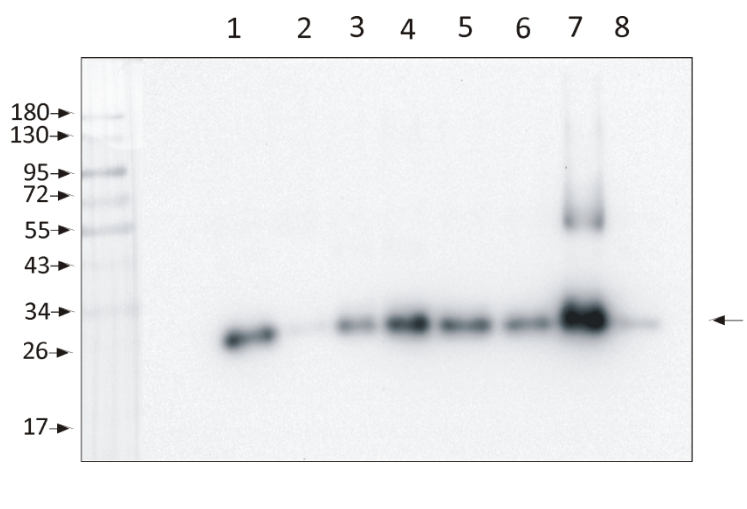
1,5 µg of chlorophyll from Zea mays, mesophyll (1) and bundle sheath (2), Panicum miliaceum, mesophyll (3) and bundle sheath (4), Echinochloa crus-galli, mesophyll (5) and bundle sheath (6), Arabidopsis thaliana (7), Pisum sativum (8) chloroplasts extracted with 0.4 M sorbitol, 50 mM HEPES NaOH, pH 7.8, 10 mM NaCl, 5 mM MgCl2 and 2 mM EDTA were loaded to lanes. Samples were denatured with Laemmli buffer at 75 0C for 5 min and were separated on 12% SDS-PAGE, and blotted 30 min to PVDF using wet transfer. Blot was blocked with 5% BSA for 2h at room temperature (RT) with agitation. Blot was incubated in the primary antibody Anti-Lhcb1-P (LOT 2201) at a dilution of 1: 10 000 in 1% BSA in TBS-T overnight at 40C with agitation. The antibody solution was decanted, and the blot was washed 4 times for 5 min in TBS-T at RT with agitation. Blot was incubated in secondary antibody (anti-rabbit IgG HRP conjugated, from Agrisera, AS09 602, LOT 2106) diluted to 1: 20 000 in 1 % BSA in TBS-T for 1h at RT with agitation. The blot was washed 5 times for 5 min in TBS-T and 2 times for 5 min in TBS, and developed for 1 min with 1.25 mM luminol, 0.198 mM coumaric acid and 0.009% H2O2 in 0.1 M Tris- HCl, pH 8.5. Exposure time in ChemiDoc System was 153 seconds.Courtesy Dr. Wioleta Wasilewska-Dębowska, Warsaw University, Poland
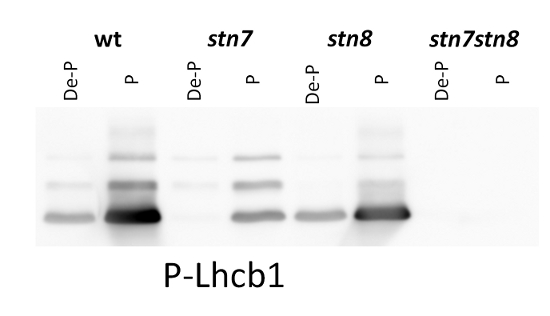
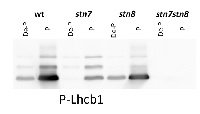
1 µg of thylakoid membranes isolated from Arabidopsis thaliana wilde-type and mutants were solubilized with 3X LB (6 M urea, 12% SDS, 30% glycerol, 100 mM DTT, 150 mM Tris pH7.0, 0.8% Comassie G-250). 1 µg of total chlorophyll was loaded and separated on 16% SDS-PAGE, and then blotted for 2 h onto nitrocellulose membrane. Blots were blocked with milk powder for 2 h and then incubated in the primary antibody solution, at a dilution of 1: 5 000/2.5 h, RT incubation, which was then decanted and the blot was washed 3 times for 5 min in TBST. Membrane was incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated) diluted to 1:10 000 in for 1h, followed by washing steps as above. All the steps fallowing transfer were performed in room temperature (RT) with agitation. Membrane was developed for 5 min with ECL according to the manufacturer’s instructions and recorded using FujiFilm CCD camera with 30 s increment time for around 5 min.
Courtesy of a PhD candidate Małgorzata Pietrzykowska, Umeå Plant Science Centre, Sweden - Background
-
Background: The major light-harvesting antenna complex II (LHCII) in photosynthetic eukaryotes is located in the thylakoid membrane of the chloroplast. It is a heterotrimeric complex formed by up to 3 different individual subtypes of chlorophyll a/b-binding proteins: Lhcb1, Lhcb2, and Lhcb3. Lhcb1 is the most abundant chlorophyll a/b-binding protein in eukaryotic phototrophs and often is coded by several nuclear genes.
- Product Citations
-
Selected references: Nilsson et al. (2020). PSB33 protein sustains Photosystem II in plant chloroplasts under UVA light. J Exp Bot. 2020 Sep 15;eraa427.doi: 10.1093/jxb/eraa427.
Rudenko et al. (2019). The role of carbonic anhydrase a-CA4 in the adaptive reactions of photosynthetic apparatus: the study with a-CA4 knockout plants. Protoplasma (2019). https://doi.org/10.1007/s00709-019-01456-1
Rantala and Tikkanen et al. (2018). Phosphorylation-induced lateral rearrangements of thylakoid protein complexes upon light acclimation. Plant Direct Vol. 2, Issue 2.
Rantala et al. (2017). Proteomic characterization of hierarchical megacomplex formation in Arabidopsis thylakoid membrane. Plant J. 2017 Dec;92(5):951-962. doi: 10.1111/tpj.13732.
Fristedt et al. (2017). PSB33 sustains photosystem II D1 protein under fluctuating light conditions. Journal of Experimental Botany doi:10.1093/jxb/erx218. - Protocols
-
- Reviews:
-
Soo Yeon Ko | 2020-11-18We always use this antibody when we check the Lhcb1-p band in Western blot (1:10000 dilution) . It is also working on Oriza Sativa. We used it with isolated 2ug thylakoid membrane.Soo Yeon Ko | 2019-11-18The antibody is working on Oriza Sativa and Arabidopsis thaliana. We used it with isolated 2ug thylakoid membrane and could get the band (1:5000 dilution)Vasily Terentyev | 2017-11-29These antibodies worked very well with thylacoid preparations from Arabidopsis and Barley. Only one spesific thing should be noted about the protocol is that blotting with the primary antibody should be continued overhight at 4.Stefano Caffarri | 2016-09-22Very sensitive and specific antibody for P-Lhcb1. See also Crepin A et al. BBA 2015
Accessories

AS01 004 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Photosynthetic eukaryotes including A. thaliana, A. hypogaea, B. napus, C.sinensis, C. reticulata, C. vulgaris, C. quitensis Kunt Bartl, C. pumilum, H. vulgare, L. esculentum (Solanum lycopersicon), M. crystallinum, N. tabacum, O. sativa, P. sativum, P. vulgaris, R. discolor, S. alaba, S. vulgaris, S. lycopersicum, S. oleracea, T. aestivum, Z. mays


