1
Fucose
AS07 268 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Higher plants
- Product Info
-
Immunogen: Core fucose residues bound to the N-glycan in alpha 1,3
Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: ELISA (ELISA), Immunolocalization (IL), Western blot (WB) Recommended dilution: 0,5 µg/ml (ELISA), 1 : 40 (IL), 1 ug/10 ml (WB) Expected | apparent MW: 10 - 100 for various glycoproteins
- Reactivity
-
Confirmed reactivity: Higher plants, Pheodactylum tricornutum (diatom) Predicted reactivity: Higher plants, Phaeodactylum tricornutum Not reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
Western blot
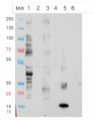
The Anti-fucose antibodies exhibit a positive response with PLA2 and Avidin and a negative one with asialofetuin. This suggests this antibody recognizes only α1,3 fucose epitopes.
For each sample, 1µg of purified protein was denatured with leammli sample buffer at 100°C for 10 minutes loaded on a Bis-Tris gel 4-12% and blotted 1h15 to nitrocellulose membrane using semi-dry transfer. Blots were saturated in TBST over night with agitation. Blot was incubated for 2 hours with primary antibody at a dilution of 1:5000 in TBST at RT with agitation. The antibody solution was discarded and after quick whashes with TBST, the blot was washed 3X15 min in TBST. Blot was incubated in secondary antibody (Goat anti Rabbit IgG, HRP conjugated AS09 602 lot 1707 from AGRISERA) diluted to 1:50 000 in TBST for 1 hour at RT with agitation. The blot was washed as above, plus 1 wash in TBS for 10 min at RT with agitation. The blot was developed for 5 min with chemiluminescent detection reagent. Exposure time was 5 seconds.
RiboB: RiboNuclease B contains only oligomannosidic N-Glycans. Used here as a negative control. PLA2: Phospholipase A2 from honey bee venom (Apis mellifera) contains α1,3 Fucose residues. aFet: Asialofetuin is a glycoprotein with α1,6 fucose. Used here as a negative control. Av: recombinant Avidin expressed in corn contains α1,3 Fucose residues.
Courtesy Dr. Muriel Bardor, University of Rouen, France
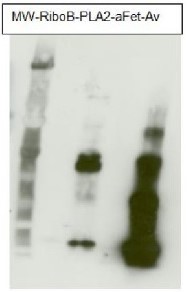
Total cell extract from Arabidopsis thaliana wild type (1) and cell extracts from different mutants defective in fucosyltransferases (2-5) (data not published yet).
Primary antibody has been used at 10 µg/10 ml of incubation buffer. Detection has been done using enchanced chemiluminescence (ECL).
Dot blot reaction of anti-Fucose and anti-Xylose antibodies with various controls: Avidin (Fuc+/Xyl+), Fetuin (Fuc-/Xyl-), PLA2 (Fuc+/Xyl-) and Mur1-2 (Fuc-/Xyl+). 2 µl of each extract were spotted on a nitrocellulose membrane, placed on top of 2 WHATMAN filters (one soaked in TBS-T) and dried for 1.5 h at RT. The mem-brane was blocked for 30 min with 2% low-fat milk powder in TBS-T (0.1% TWEEN 20) and incubated with anti-Fucose(1) (AS07 268, 1:1000) or anti-Xylose(2) (AS07 267, 1:1000) for 30 min and then with secondary anti-rabbit(1:1000) antibody (ALP conjugated, recommended secondary antibody AS09 607). Membrane was washed with TBS-T 3 x 10 minutes before reaction development using alkaline phosphatase reagent BCIP®/NTB premixed solution (Sigma, Prod. No. B6404).
Please follow this link for a more detailed Dot-Blot protocol
Immunolocalization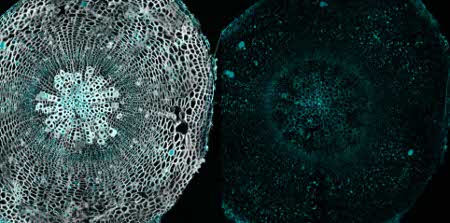
Immunolocalization using anti-fucose antibodies on a 40µm vibratome section of a 21 day old Arabidopsis thaliana hypocotyl (wood). Fixation: 4% formaldehyde fixation in 1X PME (100 mM PIPES, 1 mM MgSO4, 2 mM EGTA); blocking: 5 % BSA; primary antibody dilution: 1:40; secondary antibody: 1:300 AlexaFluor568 from Invitrogen (Life Technologies), Alexa Fluor® 568 Goat Anti-Rabbit IgG (H+L); Left panel, immunolabel channel (cyan) overlaid on grayscale channel (0.001% Calcofluor white which stains cellulose and hemicellulose). Right panel, only immunolabelling
Courtesy of Dr. Hardy Hall and Dr. Urs Fischer, Umeå Plant Science Centre, SwedenApplication examples: 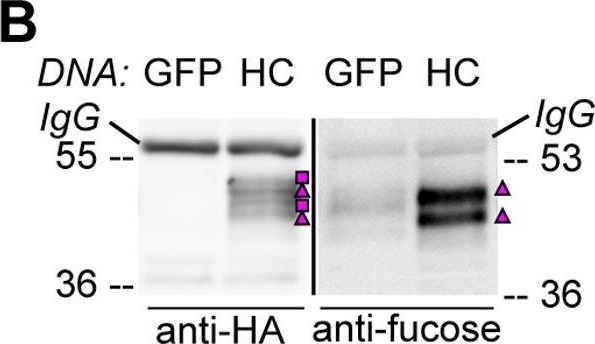
Reactant: Arabidopsis thaliana (Thale cress)
Application: Western Blotting
Pudmed ID: 21695217
Journal: PLoS One
Figure Number: 3B
Published Date: 2011-06-23
First Author: Burén, S., Ortega-Villasante, C., et al.
Impact Factor: 2.942
Open PublicationHA-tagged CAH1 contains both high mannose-type and complex-type N-glycans.Analysis of protein extracts of protoplasts from Arabidopsis suspension culture cells transiently expressing GFP (negative control), HA-tagged wt (HC) or single mutated (N4) CAH1. Heavy chain from immunoprecipitation is indicated as IgG. (A) HC protein migrates as four distinct bands: two isoforms are sensitive (squares) and two resistant (triangles) to Endo H. Deglycosylated protein is labelled with a dot. (B) HA-immunoprecipitation of HC reveals four protein isoforms when probed with HA antibodies, (squares and triangles) two of which are recognized by ?(1,3)-fucose antibodies (triangles). (C) Endo H treatment of HC shows that the fucose containing glycoforms are resistant to the enzyme (triangles). The glycoforms detected by Con A affinoblot (squares) are sensitive to Endo H, corresponding to high mannose-type glycoforms. (D) HC migrates as two main bands when expressed in Columbia (Col)-0 mesophyll protoplasts from the cgl mutant, which is unable to produce complex-type N-glycans. (E) HA immunoprecipitation of HC reveals four protein isoforms, while only two can be detected of the N4 mutant using HA antibodies (triangles and squares). Analysis using ?(1,3)-fucose antibodies on the same samples showed that only two of the HC, and one of the N4, glycoforms harbour complex-type glycans (triangles). (F) Endo H treatment and HA immunodetection of total protein extract confirmed that only one glycoform in N4 and two in HC are resistant to the enzyme (triangles). Deglycosylated isoforms are marked with dots.
Reactant: Arabidopsis thaliana (Thale cress)
Application: Western Blotting
Pudmed ID: 21695217
Journal: PLoS One
Figure Number: 3C
Published Date: 2011-06-23
First Author: Burén, S., Ortega-Villasante, C., et al.
Impact Factor: 2.942
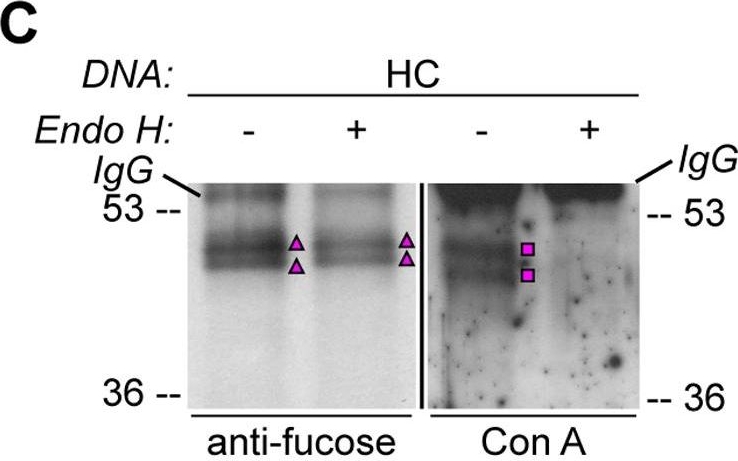
Open PublicationHA-tagged CAH1 contains both high mannose-type and complex-type N-glycans.Analysis of protein extracts of protoplasts from Arabidopsis suspension culture cells transiently expressing GFP (negative control), HA-tagged wt (HC) or single mutated (N4) CAH1. Heavy chain from immunoprecipitation is indicated as IgG. (A) HC protein migrates as four distinct bands: two isoforms are sensitive (squares) and two resistant (triangles) to Endo H. Deglycosylated protein is labelled with a dot. (B) HA-immunoprecipitation of HC reveals four protein isoforms when probed with HA antibodies, (squares and triangles) two of which are recognized by ?(1,3)-fucose antibodies (triangles). (C) Endo H treatment of HC shows that the fucose containing glycoforms are resistant to the enzyme (triangles). The glycoforms detected by Con A affinoblot (squares) are sensitive to Endo H, corresponding to high mannose-type glycoforms. (D) HC migrates as two main bands when expressed in Columbia (Col)-0 mesophyll protoplasts from the cgl mutant, which is unable to produce complex-type N-glycans. (E) HA immunoprecipitation of HC reveals four protein isoforms, while only two can be detected of the N4 mutant using HA antibodies (triangles and squares). Analysis using ?(1,3)-fucose antibodies on the same samples showed that only two of the HC, and one of the N4, glycoforms harbour complex-type glycans (triangles). (F) Endo H treatment and HA immunodetection of total protein extract confirmed that only one glycoform in N4 and two in HC are resistant to the enzyme (triangles). Deglycosylated isoforms are marked with dots.
Reactant: Arabidopsis thaliana (Thale cress)
Application: Western Blotting
Pudmed ID: 21695217
Journal: PLoS One
Figure Number: 3E
Published Date: 2011-06-23
First Author: Burén, S., Ortega-Villasante, C., et al.
Impact Factor: 2.942
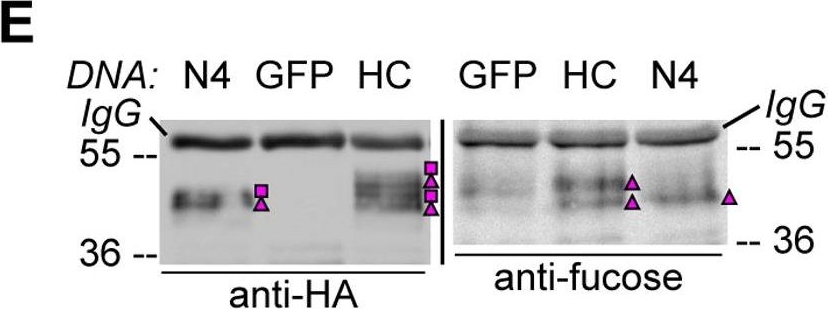
Open PublicationHA-tagged CAH1 contains both high mannose-type and complex-type N-glycans.Analysis of protein extracts of protoplasts from Arabidopsis suspension culture cells transiently expressing GFP (negative control), HA-tagged wt (HC) or single mutated (N4) CAH1. Heavy chain from immunoprecipitation is indicated as IgG. (A) HC protein migrates as four distinct bands: two isoforms are sensitive (squares) and two resistant (triangles) to Endo H. Deglycosylated protein is labelled with a dot. (B) HA-immunoprecipitation of HC reveals four protein isoforms when probed with HA antibodies, (squares and triangles) two of which are recognized by ?(1,3)-fucose antibodies (triangles). (C) Endo H treatment of HC shows that the fucose containing glycoforms are resistant to the enzyme (triangles). The glycoforms detected by Con A affinoblot (squares) are sensitive to Endo H, corresponding to high mannose-type glycoforms. (D) HC migrates as two main bands when expressed in Columbia (Col)-0 mesophyll protoplasts from the cgl mutant, which is unable to produce complex-type N-glycans. (E) HA immunoprecipitation of HC reveals four protein isoforms, while only two can be detected of the N4 mutant using HA antibodies (triangles and squares). Analysis using ?(1,3)-fucose antibodies on the same samples showed that only two of the HC, and one of the N4, glycoforms harbour complex-type glycans (triangles). (F) Endo H treatment and HA immunodetection of total protein extract confirmed that only one glycoform in N4 and two in HC are resistant to the enzyme (triangles). Deglycosylated isoforms are marked with dots.
Reactant: Arabidopsis thaliana (Thale cress)
Application: Western Blotting
Pudmed ID: 21695217
Journal: PLoS One
Figure Number: 4B
Published Date: 2011-06-23
First Author: Burén, S., Ortega-Villasante, C., et al.
Impact Factor: 2.942
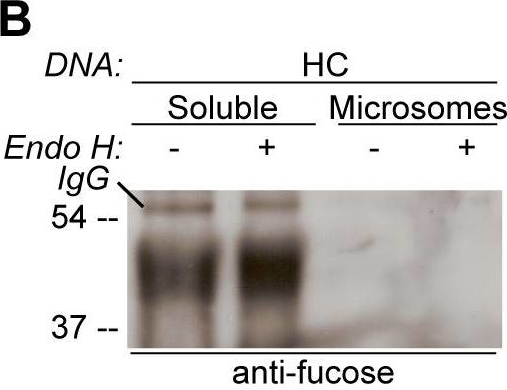
Open PublicationGlycosylation state of CAH1 found in the chloroplast stroma containing fraction is different than for microsome associated CAH1.Analysis of soluble (chloroplast stroma-containing) and microsome fractions of an Arabidopsis cell suspension culture stably expressing HC protein. Heavy chain from immunoprecipitation is indicated as IgG. (A) HA-immunoprecipitated HC from the soluble fraction is mainly resistant to Endo H (triangle), while most of HC from the microsome fraction was sensitive to Endo H (dot). (B) Only HA-immunoprecipitated protein from the soluble fraction can be detected using ?(1,3)-fucose antibodies. (C) Schematic visualization of the proposed glycoforms of epitope-tagged CAH1 (blue, yellow, orange, grey and red squares and triangles symbolize N-glycosylation site 1–5, respectively). High-mannose type glycans are showed as squares and complex-type glycans as triangles.
- Additional Information
-
Additional information: Alpha (1,3) fucose is present not only in plants but also in some invertebrates (such as nematodes, bees, etc,) , However, cross-reaction with glycoproteins from these organisms is weaker than the one observed in plants, This sugar residue does not exist in mammals, in their endogenous glycoproteins Additional information (application): Controls:
PLA2 (phospholipase 2 from bee venom) which contains only α1.3 fucose, Sigma, product number P9279.
Type II - horseradish peroxidase which contains β1.2 Xylose and α1.3 fucose, Sigma, product number P8250.
The antibody does not recognize alpha 1,6-fucose.
- Background
-
Background: This antibody specifically cross-reacts against fucose residues bound to the protein N-glycans in alpha 1,3. This residue is characteristic of the plant protein N-glycans and is absent in protein N-glycans from animals. This residue is added in the Golgi apparatus.
- Product Citations
-
Selected references: Xu et al. (2023). The Golgi-localized transporter OsPML4 contributes to manganese homeostasis in rice. Plant Sci. 2023 Dec 2:339:111935.doi: 10.1016/j.plantsci.2023.111935.
Zhang et al. (2018). An important role of L -fucose biosynthesis and protein fucosylation genes in Arabidopsis immunity. New Phytol. 2018 Dec 15. doi: 10.1111/nph.15639.
Jansing et al. (2018). CRISPR/Cas9-mediated knockout of six glycosyltransferase genes in Nicotiana benthamiana for the production of recombinant proteins lacking β-1,2-xylose and core α-1,3-fucose. Plant Biotechnol J. 2018 Jul 3. doi: 10.1111/pbi.12981.
Nakanishi et al. (2017). Protection of Human Colon Cells from Shiga Toxin by Plant-based Recombinant Secretory IgA. Sci Rep. 2017 Apr 3;7:45843. doi: 10.1038/srep45843. (ELISA)
Hanania et al. (2017). Establishment of a tobacco BY2 cell line devoid of plant specific xylose and fucose as a platform for the production of biotherapeutic proteins. Plant Biotechnol J. 2017 Feb 3. doi: 10.1111/pbi.12702.
Ebert et al. (2015). Identification and Characterization of a Golgi-Localized UDP-Xylose Transporter Family from Arabidopsis. Plant Cell. 2015 Mar 24. pii: tpc.114.133827.
Lehtimäki et al. (2014). Posttranslational modifications of FERREDOXIN-NADP+ OXIDOREDUCTASE in Arabidopsis chloroplasts. Plant Physiol. 2014 Dec;166(4):1764-76. doi: 10.1104/pp.114.249094
Baiet et all. (2010). N-glycans of Phaeodactylum tricornutum diatom and functional characterization of its N-acetylglucosaminyltransferase I enzyme. J Biol. Chem. Dec 17. - Protocols
- Agrisera Western Blot protocol and video tutorials
- Reviews:
-
This product doesn't have any reviews.