1
AtpH | ATP synthase subunit c (chloroplastic)
AS09 591 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, C. reinhardtii
- Product Info
-
Immunogen: KLH-conjugated peptides derived from AtpH subunit c of Arabidopsis thaliana UniProt: P56760, TAIR: AtCg00140 and Chlamydomonas reinhardtii UniProt: Q37304
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 100 �l Reconstitution: For reconstitution add 100 �l of sterile water Storage: Store lyophilized/reconstituted at -20�C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 10 000 (WB) Expected | apparent MW: 8 kDa (for Arabidopsis thaliana)
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Chlamydomonas reinhardtii Predicted reactivity: Algae, Cannabis sativa, Glycine max, Hordeum vulgare, Oryza sativa, Ostreococcus tauri, Physcomitrium patens, Pinus thunbergii, Pisum sativum, Populus alba, Zea mays, Vitis vinifera
Species of your interest not listed? Contact usNot reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
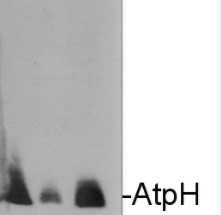
Application example

10 ug of chlorophyll/well of Chlamydomonas reinhardtii total cell extract (1), Chlamydomonas reinhardtii subunit gamma deletion mutant thylakoid membrane fraction (2), Arabidospsis thaliana thylakoid membrane fraction (3), Chlamydomonas reinhardtii thylakoid membrane preparation (4) were separated on 12-18% acrylamide-8M urea gel and blotted to nitrocellulose membrane. Filters were blocked 1 h with 5% dry milk in 1 x PBS and probed with anti-ATP synthase subunit c antibody (AS09 591, 1: 10 000, 1h) and secondary HRP-conjugated anti-rabbit antibody (1: 10 000, 1 h) in 1 x PBS containing 5% dry milk. All steps were performed at RT with agitation. Signal was detected with chemiluminescent detection, exposure time 30’’.
Arabidopsis membrane preparation has been done according to Lezhneva et al. (2008) A novel pathway of cytochrome c biogenesis is involved in the assembly of the cytochrome b6f complex in arabidopsis chloroplasts. J Biol. Chem., 283:24608-24616 and Chlamydomonas membranes were prepared according to Chua & Bennoun (1975) Thylakoid membrane polypeptides of Chlamydomonas reinhardtii: wild-type and mutant strains deficient in photosystem II reaction center. PNAS 72:2175-2179
Courtesy Dr. Yves Choquet, CNRS, France
- Additional Information
-
Additional information: This product can be sold containing ProClin if requested Additional information (application): Please note that increased incubation at 95�C (20-30 min) prior to loading is recommended to break the multimeric c-mer structure, detection of partial ring structures (e,g, 5 or 6 subunits) may occur - Background
-
Background: F-type ATPase (ATP synthase) is the universal enzyme that synthesizes ATP from ADP and phosphate using the energy stored in a transmembrane ion gradient. Multiple copies of the c subunit build up the ring structure (in spinach a 14-mer of ~112 kDa) of the membrane bound Fo-part of the enzyme.
- Product Citations
-
Selected references: Schulz et al. (2017). Molecular architecture of the N-type ATPase rotor ring from Burkholderia pseudomallei. EMBO Rep. 2017 Apr;18(4):526-535. doi: 10.15252/embr.201643374.
- Protocols
- Reviews:
-
This product doesn't have any reviews.


