1
Cas9 | CRISPR-associated endonuclease 9 (monoclonal)
AS17 4124 | Clonality: Monoclonal | Host: Mouse | Reactivity: Cas9 from Streptococcus pyogenes
- Data sheet
-
- Product Info
-
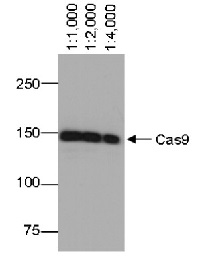
Sub class: IgG1k Immunogen: Recombinant protein part from the N-terminus of Cas9 from Streptococcus pyogenes. Host: Mouse Clonality: Monoclonal Purity: Total IgG. Protein A purified in PBS. Contains 0.02 % sodium azide. Format: Liquid Quantity: 50 �g Storage: Store lyophilized/reconstituted at -20�C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. For long term storage -80�C is recommended. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Immunofluorescence (IF), Immunoprecipitation (IP), Western blot (WB) Recommended dilution: 1 : 200 (IF), (IP), 1 : 1000-1 : 5000 (WB) Expected | apparent MW: Depends upon a MW of a protein which is a fusion partner - Reactivity
-
Confirmed reactivity: Cas9 from Streptococcus pyogenes - Application Examples
-
Application example 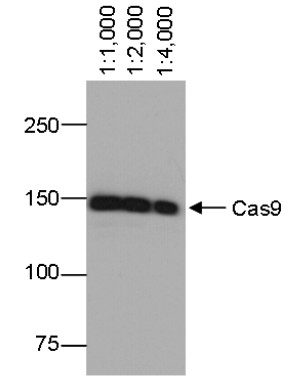
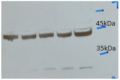
Protein extracts from HEK293 cells were transfected with a myc-tagged Cas9. Visualization was made using monoclonal anti-Cas) antibodies used in annotated dilutions.
Protein samples were separated on 7.5 % SDS-PAGE and blotted 1h to nitrocellulose membrane. Blots were blocked with 3 % non-fat dry milk in PBS+Tween for 1h at room temperature (RT) with agitation. Blot was incubated in the primary antibody at a given dilution for 1h at RT with agitation in PBS-T. The antibody solution was decanted and the blot was rinsed briefly twice, then washed 3 times for 5 min in TBS-T at RT with agitation. Blot was incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated) diluted to 1:10 000 in for 1h at RT with agitation. The blot was washed as above and incubated in ECL solution followed by exposure to an X-ray film.
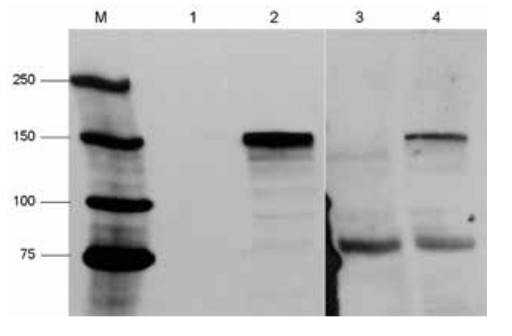 Protein extracts from HEK293 cells were transfected with a myc-tagged Cas9. Transfected cells (lane 2 and 4) and untransfected cells (lane 1 and 3) cells. Detection using anti-myc antibody (lane 3 and 4).
Protein extracts from HEK293 cells were transfected with a myc-tagged Cas9. Transfected cells (lane 2 and 4) and untransfected cells (lane 1 and 3) cells. Detection using anti-myc antibody (lane 3 and 4).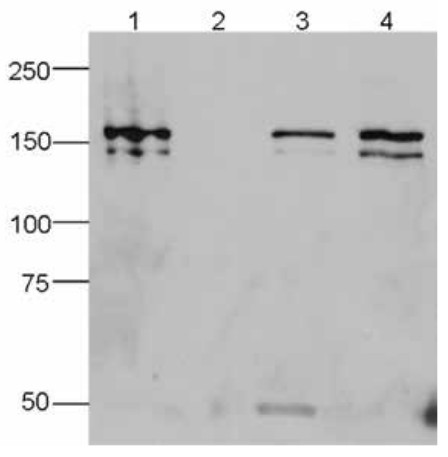
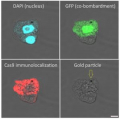
IP was performed on whole cell extracts (100 μg) from HEK293 cells transfected with a Flag-tagged Cas9 using monoclonal anti-Cas9 antibodies. The immunoprecipitated proteins were subsequently analysed by Western blot with the antibody. Lane 3 and 4 show the result of the IP; a negative IP control (IP on untransfected cells) and the input (15 μg) are shown in lane 2 and 1, respectively.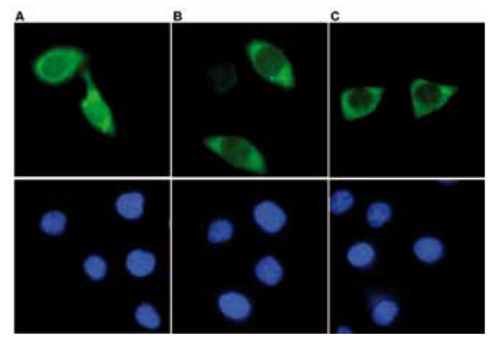 Hela cells were transiently transfected with a Flag-tagged Cas9 expression vector. 48 hours post transfection the cells were fixed in 3.7% formaldehyde, permeabilized in 0.5% Triton-X-100 and blocked in PBS containing 2% BSA for 2 hours at RT. The cells were stained with monoclonal anti-Cas9 (A and B) or with an anti- Flag (C) antibody at 4°C O/N, followed by incubation with an anti-mouse secondary antibody coupled to AF488 for 1 h at RT. Nuclei were counter-stained with Hoechst 33342 (bottom).
Hela cells were transiently transfected with a Flag-tagged Cas9 expression vector. 48 hours post transfection the cells were fixed in 3.7% formaldehyde, permeabilized in 0.5% Triton-X-100 and blocked in PBS containing 2% BSA for 2 hours at RT. The cells were stained with monoclonal anti-Cas9 (A and B) or with an anti- Flag (C) antibody at 4°C O/N, followed by incubation with an anti-mouse secondary antibody coupled to AF488 for 1 h at RT. Nuclei were counter-stained with Hoechst 33342 (bottom). - Background
-
Background: Cas9 (CRISPR-associated endonuclease 9) is an RNA-guided DNA endonuclease associated with the Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) type II adaptive immunity system. CRISPR clusters are transcribed and processed into CRISPR RNA (crRNA). Cas9 protein serves as a genome engineering tool to induce site-directed double strand breaks in DNA. Alternative name: Csn1. - Protocols
-
- Reviews:
-
This product doesn't have any reviews.