1
Dehydrin (affinity purified serum)
AS07 206A | Clonality: Polyclonal | Host: Rabbit | Reactivity: Higher plants
- Product Info
-
Immunogen: KLH-conjugated peptide sequence TGEKKGIMDKIKEKLPGQH of K-segment conserved in a wide range of plant species
Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile, deionized water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 1000 (WB) Expected | apparent MW: 9-200 kDa
- Reactivity
-
Confirmed reactivity: Brassica oleracea, Cucumis sativus, Lycopersicon esculentum, Pinus sylvestris, Pistacia vera Predicted reactivity: Glycine max, Hordeum vulgare, Nicotiana tabacum, Oryza sativa, Pisum sativum, Populus sp., Zea mays
Species of your interest not listed? Contact usNot reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
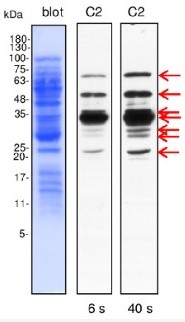
Application example 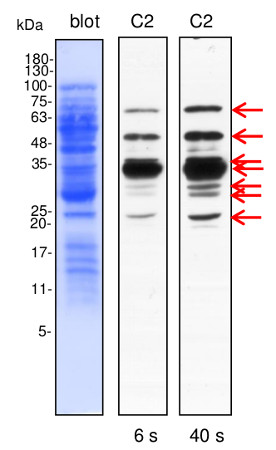
Line C2- 10 µg of Brassica oleracea mitochondrial proteins (C2- plants grown in severe drought conditions) isolated as described by Rurek et al., 2015 (doi: 10.1016/j.bbabio.2015.01.005) were separated by 12% SDS-PAGE and electroblotted in semi-dry conditions (Towbin buffer) to Immobilon-P membrane (Millipore). Blots were CBB R 250 briefly stained, destained, wet-scanned and after completed destaining, they were blocked in 5% skimmed milk (dissolved in PBS-T containing 0.1% Tween 20) in 1h, RT. Primary antisera (at 1: 1000, diluted in 2% skimmed milk in PBS-T) were bound by overnight incubation of blots at 4°C. After blot washing (2 times quick, 2 times of 5 min, and 10 min at the end), secondary goat anti-rabbit IgGs, HRP- conjugated (Agrisera product AS09 602; at 1: 50000, diluted in 2% milk/ PBS-T) were bound in 1 h, RT. Blots were washed (as above) with copious amounts of PBS-T and chemiluminescence signals acquired by using standard ECL reagents on RTG film between 3 s and 2 min (periods of the given image acquisition were indicated).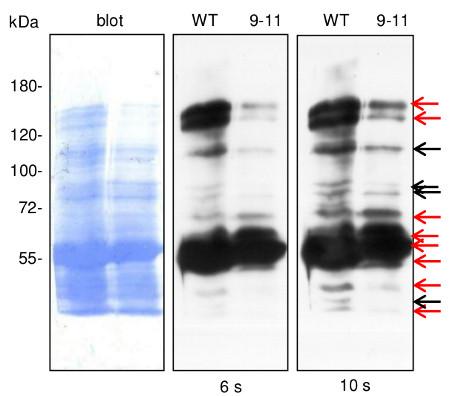
In the nuclear fraction of Arabidopsis thaliana, few HMW proteins immunoreactive with anti-dehydrin anstiserum were also detected. The differences between their abundance in WT and 9-11 lines could be, however, ascribed to the actual protein loading on the blot. Experimental procedure was conducted as above.Lines WT and 9-11- ca. 20 µg of proteins enriched in nuclear fraction from Arabidopsis thaliana leaf rosettes (WT- wild type; 9-11- T-DNA insertion mutants for h2a.z gene encoding particular histon H2A.Z isoform) were resolved by SDS-PAGE and HMW (high-molecular weight) proteins immunoreactive with dehydrin antisera were analysed. After separation, proteins were electroblotted and immunodetected essentially in the same manner as it was indicated above.
Application of WT/9-11 blot was possible due to the courtesy of the Department of Biotechnology (Institute of Molecular Biology and Biotechnology, Faculty of Biology, Adam Mickiewicz University in Poznań). All remaining blots as well as all immunodetection assays were prepared by Dr Michał Rurek (Department of Molecular and Cellular Biology of the same Institute).
- Background
-
Background: Dehydrins are stress proteins involved in formation of plant protective reactions against dehydration. They are normally synthesized in maturating seeds during their dessication, as well as in vegetative tissues of plants treated with abscisic acid or exposed to environmental stress factors that result in cellular dehydration.
- Product Citations
-
Selected references: Hernandez et al. (2023). Tissue-specific modulation of functional stress responsive proteins and organic osmolytes by a single or dual short-term high CO2 treatment during long-term cold storage Autumn Royal table grapes. Postharvest Biology and Technology Volume 205, November 2023, 112501
ZamaniBahramabadi et al. (2019). Dehydrin Content in Fresh and Desiccated Pistachio (Pistacia vera L.) Seeds. Iranian Journal of Science and Technology, Transactions A: Science, pp 1–7.
Szegő et al. (2019). Diverse responsiveness of dehydrin genes to abscisic acid and water stress treatments in cucumber F1 cultivar hybrids. The J of Horticult Science and Biotechnology. June 2019.
Goñi et al. (2018). Ascophyllum nodosum extract biostimulants and their role in enhancing tolerance to drought stress in tomato plants. Plant Physiol Biochem. 2018 May;126:63-73. doi: 10.1016/j.plaphy.2018.02.024. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts - Reviews:
-
This product doesn't have any reviews.




