1
PsaA | PSI-A core protein of photosystem I
AS06 172 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, C. quitensis Kunt Bartl, C. pumilum, C. reinhardtii, C. zofingiensis, F. vesiculosus, H.vulgare, M. polymorpha, N. oceanica, N. tabacum, O. sativa, P. abies, P. sativum, P. strobus, P. vulgaris, S. oleracea, C.reinhardtii, Synechococcus PCC 7942, Synechocystis PCC 6803, Scenedesmus obliquus, microalgae N. gaditana
- Product Info
-
Immunogen: N-terminal part of recombinant PsaA protein from Chlamydomonas reinhardtii P12154
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Blue Native PAGE (BN-PAGE), Immunogold (IG), Western blot (WB) Recommended dilution: 1 : 20 (IG), 1 : 1000-1 : 5000 (WB) Expected | apparent MW: 82 | 55-60 kDa
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Begonia sp. , Bryopsis corticulans, Chlamydomonas reinhardtii, psychrophilic Chlamydomonas sp. UWO241 and Chlamydomonas sp. ICE-MDV, Chlorella sorokiniana, Chlorella vulgaris, Chromochloris zofingiensis, Colobanthus quitensis Kunt Bartl, Craterostigma pumilum, Cytisus cantabricus (Wilk.) Rchb. F., Dianthus caryophyllus, Dioxoniella giordanoi (red alga), Drosera capensis, Euonymus japonicus, Fraxinus ornus, Fucus vesiculosus, Haematococcus pluvialis, Halomicronema hongdechloris, Hieracium pilosella L., Hordeum vulgare, Lasallia hispanica, Nannochloropsis oceanica strain IMET1, Nicotiana benthamiana, Nicotiana tabacum, Oryza sativa, Pisum sativum, Marchantia polymorpha (liverwort), micro Nannochloropsis gaditana, Phaseolus vulgaris, Physcomitrium patens, Picea abies, Pinus strobus, Sinapsis alba, Spinacia oleracea, Synechococcus PCC 7942, Synechocystis PCC 6803, Syntrichia muralis (Hedw.) Raab, Scenedesmus obliquus, Tillandsia flabellate, Ulva prolifera Predicted reactivity: Algae, Bigelowiella natans, Cannabis sativa, Catalpa bungei, Citrus x limon, Cyanobacteria, Cyanidioschyzon merolae strain 10D, Galdieria sulphuraria, Lycopersicum esculentum, Panax ginseng, Picea spinulosa, Pinus thunbergii, Phaeodactylum tricornutum, Populus alba, Thermosynechococcus elongatus (strain BP-1), Triticum aestivum
Species of your interest not listed? Contact usNot reactive in: Chromera velia
- Application Examples
-
Application example
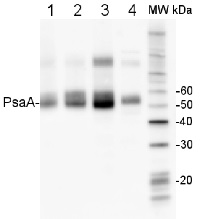
2 µg of total protein from (1) Arabidopsis thaliana leaf, (2) Hordeum vulgare leaf, (3) Chlamydomonas reinhardtii total cell, (4) Synechococcus sp. 7942 total cell all extracted with Protein Extration Buffer, PEB (AS08 300), were separated on 4-12% NuPage (Invitrogen) LDS-PAGE and blotted 1h to PVDF. Blots were blocked immediately following transfer in 2% blocking reagent in 20 mM Tris, 137 mM sodium chloride pH 7.6 with 0.1% (v/v) Tween-20 (TBS-T) for 1h at room temperature with agitation. Blots were incubated in the primary antibody at a dilution of 1: 10 000 for 1h at room temperature with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in TBS-T at room temperature with agitation. Blots were incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated, frecommended secondary antibody AS09 602) diluted to 1:50 000 in 2% blocking solution for 1h at room temperature with agitation. The blots were washed as above and developed for 5 min with chemiluminescence detection reagent according the manufacturers instructions. Images of the blots were obtained using a CCD imager (FluorSMax, Bio-Rad) and Quantity One software (Bio-Rad). Exposure time was 10 seconds.Application examples: 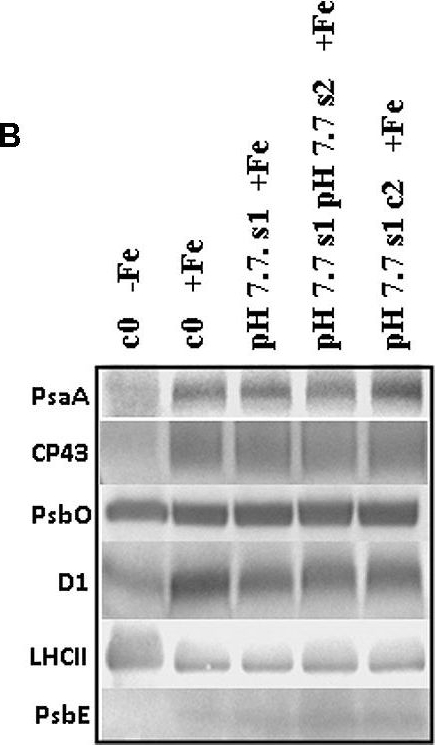
Reactant: Arabidopsis thaliana (Thale cress)
Application: Western Blotting
Pudmed ID: 26442058
Journal: Front Plant Sci
Figure Number: 6B
Published Date: 2015-10-07
First Author: Murgia, I., Giacometti, S., et al.
Impact Factor: 5.435
Open PublicationO2 evolution and protein profile of photosynthetic apparatus in A. thaliana seedlings with generational exposure to Fe deficiency. (A) SHR-trap 1445 c0, pH 7.7 s1 pH 7.7 s2, pH 7.7 s1 c2 seedlings were grown for 11 days in control AIS medium and net O2 evolution (expressed as ?mol O2 evolved min-1 mg chlorophyll-1) was measured under illumination at either 100 or 800 ?E m-2 s-1. Values are mean ± SE of three biological replicas, each consisting of at least 15 seedlings. Letters represent statistical differences, according to Student’s t-test, with p < 0.05. (B) Western blot analysis with antibodies against PsaA, CP43, PsbO, D1, LHCII, and PsbE of thylakoid membranes purified from 11 days-old SHR-trap 1445 c0 seedlings germinated in -Fe AIS medium or from 11 days-old SHR-trap 1445 c0, pH 7.7 s1, pH 7.7 s1 pH 7.7 s2, pH 7.7 s1 c2 seedlings germinated in control (+Fe) AIS medium. Samples corresponding to 1 ?g chlorophyll were loaded on each lane.
Reactant: Plant
Application: Western Blotting
Pudmed ID: 27590049
Journal: BMC Plant Biol
Figure Number: 9A
Published Date: 2016-09-02
First Author: Mazur, R., Sadowska, M., et al.
Impact Factor: 4.142
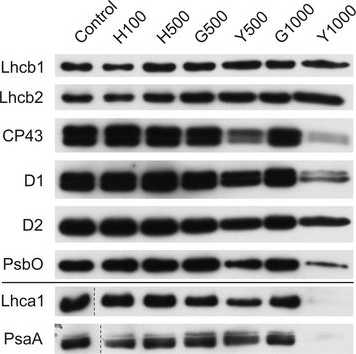
Open PublicationChanges of PSII and PSI antenna and core protein levels. Proteins from control and Tl-treated white mustard leaves were separated by SDS-PAGE followed by immunodetection with antibodies against Lhcb1, Lhcb2, Lhca1 (antenna proteins) and D1, D2, CP43, PsbO, PsaA (core proteins). Samples were loaded on the equal amount of chlorophyll (0.25 ?g). Description of samples abbreviation as given in the legend to Fig. 3
Reactant: Nicotiana tabacum (Common tobacco)
Application: Western Blotting
Pudmed ID: 28180288
Journal: J Exp Bot
Figure Number: 5A
Published Date: 2017-02-01
First Author: Schöttler, M. A., Thiele, W., et al.
Impact Factor: 6.088
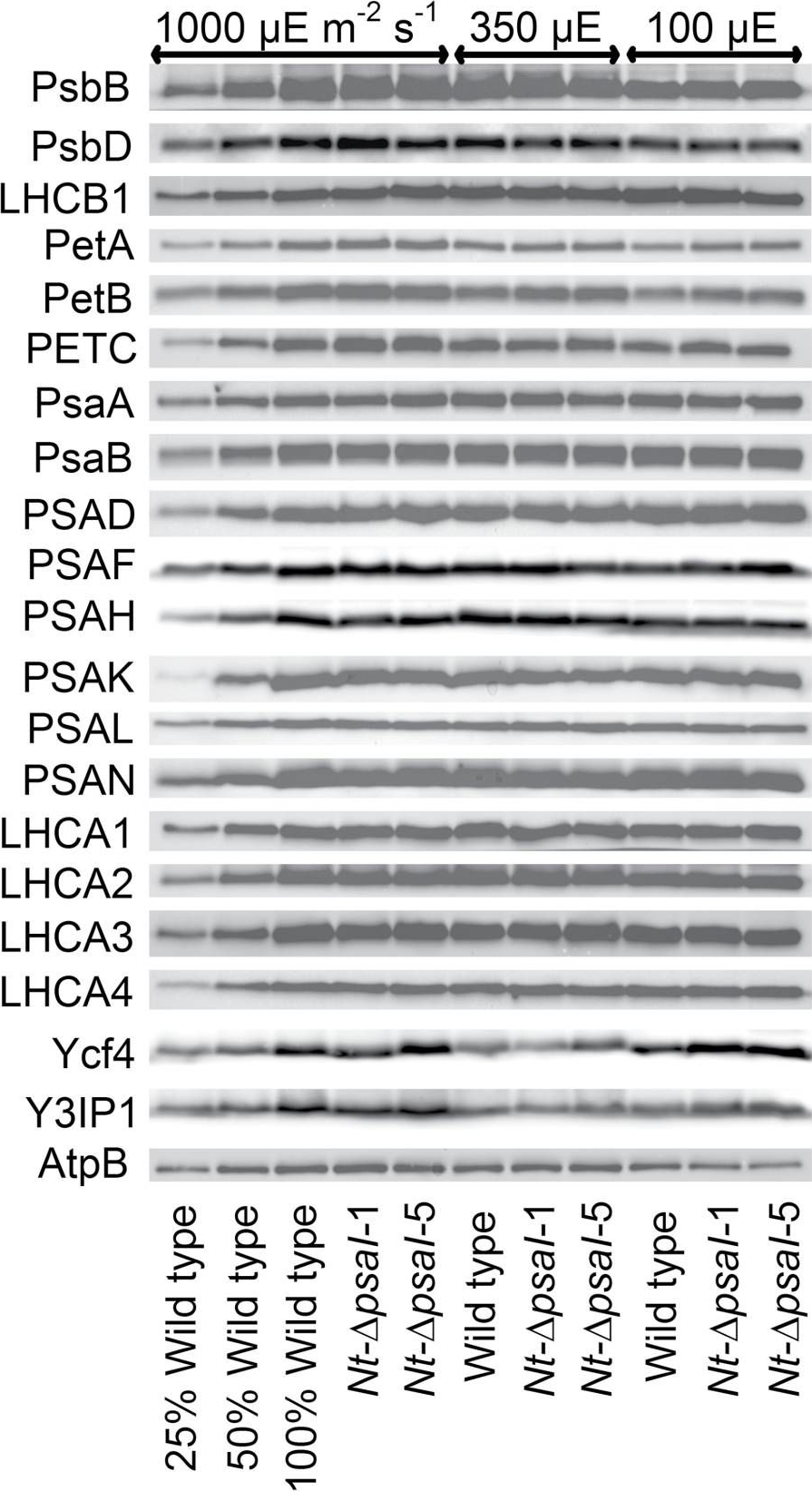
Open PublicationImmunoblot analysis of photosynthetic complex accumulation in wild-type tobacco and the two ?psaI lines grown under low, intermediate, and high-light conditions. Because the accumulation of most tested proteins was highest under high-light conditions, lanes one to three contain samples diluted to 25%, 50%, and a 100% sample of wild-type tobacco grown under high-light conditions, to allow for semi-quantitative determination of changes in protein abundance. Lanes four and five contain the two transplastomic lines grown at 1000 µE m?2 s?1. Lanes six to eight contain wild-type tobacco and the mutants grown at intermediate light intensities, and lanes nine to eleven contain samples grown at low light intensities. For PSII, the accumulation of the essential subunits PsbB (CP43) and PsbD (D2) and the LHCB1 antenna protein were determined, while for the cytochrome b6f complex, the accumulation of the essential redox-active subunits PetA (cytochrome f), PetB (cytochrome b6), and PETC (Rieske FeS protein) was tested. AtpB was probed as an essential subunit of the chloroplast ATP. For PSI, in addition to the three essential plastome-encoded subunits PsaA, PsaB, and PsaC, the accumulation of the nuclear-encoded subunits PSAD, PSAH, PSAK, PSAL, and PSAN and of the four LHCI proteins (LHCA1, LHCA2, LHCA3, LHCA4) was determined. Finally, we examined the accumulation of Ycf4, the chloroplast-encoded PSI-biogenesis factor encoded in the same operon as PsaI, and the nuclear-encoded assembly factor Y3IP1.
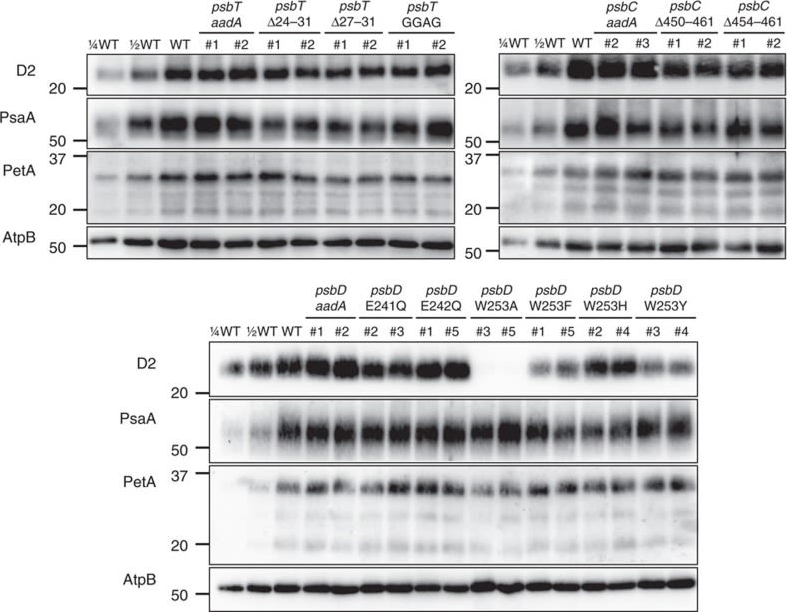
Reactant: Chlamydomonas reinhardtii (Green Alga)
Application: Western Blotting
Pudmed ID: 28466860
Journal: Nat Commun
Figure Number: 5A
Published Date: 2017-05-03
First Author: Fu, H. Y., Picot, D., et al.
Impact Factor: 13.783
Open PublicationAccumulation of photosynthetic complexes in the mutant strains.Cells were grown in TAP medium at 25?°C under LED white light (8??mol photons m?2?s?1) and collected at the mid-log phase. Two independent lines are shown for each construct. Protein samples were loaded on an equal chlorophyll basis (0.5??g per lane), and a dilution series of WT samples is shown for semi-quantitative comparison. Antibodies against essential subunits of PSII (D2), PSI (PsaA), cytochrome b6f (PetA) and ATP synthase (AtpB) probed the accumulation of the respective photosynthetic complexes. Numbers on the left side of the blots are molecular weights in kD. See Supplementary Fig. 9 for the uncropped blot images.
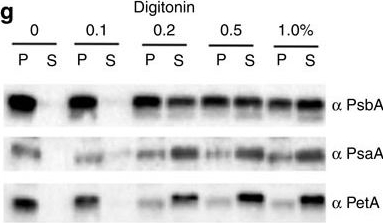
Reactant: Phaeodactylum tricornutum
Application: Western Blotting
Pudmed ID: 28631733
Journal: Nat Commun
Figure Number: 2G
Published Date: 2017-06-20
First Author: Flori, S., Jouneau, P. H., et al.
Impact Factor: 13.783
Open PublicationImmunolocalization of photosystems and of cyt b6f in the thylakoid membranes of P. tricornutum.(a–c) TEM images of P. tricornutum labelled with antibodies directed against the PsaA subunit of PSI (a), the PsbA subunit of PSII (b) and the PetA subunit of cyt b6f (c). (d) TEM micrograph of P. tricornutum thylakoid membranes showing four distinct areas: the internal membranes (‘core’: violet); the external, peripheral membranes (‘per.’: green); the pyrenoid (‘pyr.’: orange) and the envelope (‘env.’: magenta). Bars: 200?nm. (e) Principal component analysis of PSI, cyt b6f and PSII immunolocalization with the PsbA (solid squares), PsbC (open squares), PetA (cyan circle), PsaC (solid triangles) and PsaA (open triangles) antibodies. See also Supplementary Fig. 4. A total of 258 images from four independent cultures were analysed. The first two components represent more than 91% of the variance (see Supplementary Table 1, and Methods for a more detailed explanation). Green arrow: peripheral variable; violet arrow: core variable; orange arrow: pyrenoid variable; Magenta arrow: envelope variable. (f) 2D representation of the barycentre for the PSI (? PsaA+? PsaC antibodies, black square), cyt b6f (PetA, cyan circle) and PSII (? PsbA+? PsbC antibodies, red triangle) distributions. The point size along an axis is proportional to the s.d. along the corresponding component. (g) Solubilization of P. tricornutum thylakoid membranes with increasing concentrations of digitonin (0.1%, 0.2%, 0.5%, 1%). Pellet (P) and supernatant (S) were analysed by western blotting with the same anti PSI, PSII and cyt b6f antibodies as in a–c. Representative data set of an experiment replicated on three different biological samples.
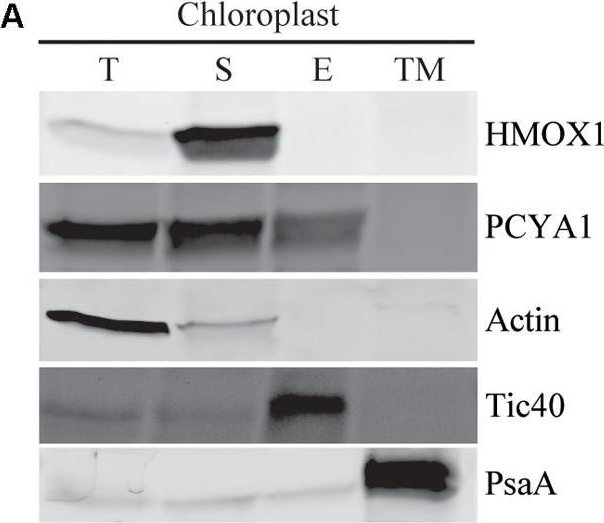
Reactant: Chlamydomonas reinhardtii (Green Alga)
Application: Western Blotting
Pudmed ID: 29875782
Journal: Front Plant Sci
Figure Number: 4A
Published Date: 2018-06-08
First Author: Zhang, W., Zhong, H., et al.
Impact Factor: 5.435
Open PublicationSubcellular distribution of CrPCYA1 in chloroplast and the export of phycocyanobilin to cytosol. (A) Biochemical fractionation of chloroplast components. Antibodies against compartmental marker proteins distributed in different chloroplast fractions were employed to distinguish the presence of CrPCYA1 via immunoblot analysis of total proteins (T), soluble fraction (S), envelope membrane (E) and thylakoid membrane (TM) from isolated chloroplast. Tic40, chloroplast envelope marker; PsaA, thylakoid membrane marker. (B) Zinc-dependent fluorescence assay. Photosensory core module (PCM) region of the prasinophyte algal DtenPHY1 purified from Chlamydomonas CC400 covalently binds phycocyanobilin (PCB). Upper panel, zinc blot; lower panel, Coomassie blue stain (CB). The two-fold serial dilutions of E. coli LMG194/pPL-PCB expressed and purified DtenPHY1 were used as positive controls. –PCB, without addition of PCB; +PCB, assembly with PCB in vitro.
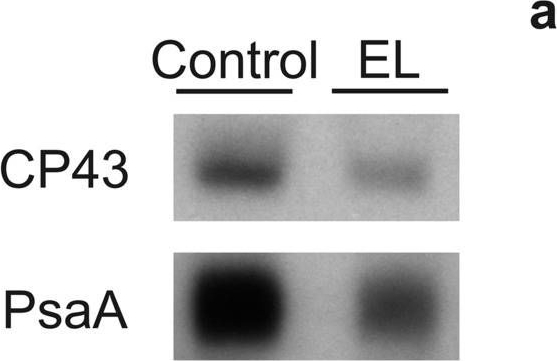
Reactant: Chlamydomonas reinhardtii (Green Alga)
Application: Western Blotting
Pudmed ID: 33280077
Journal: Photosynth Res
Figure Number: 3A
Published Date: 2021-01-01
First Author: Virtanen, O., Khorobrykh, S., et al.
Impact Factor: 3.116
Open PublicationDetection of CP43 and PsaA proteins on a film (a) and quantification of these proteins (b) from control (white bars) and EL (black bars) cells. Western blotting was done from extracted total soluble proteins, and 1 µg (CP43) or 2 µg (PsaA) of total proteins were loaded to SDS-PAGE per well. Binding of the primary and secondary antibodies was visualized via luminescence emitted by alkaline phosphatase. The signals were normalized to the average of signals originating from control samples of the respective western blot. Each bar represents an average of three biological replicates and the error bars show SD
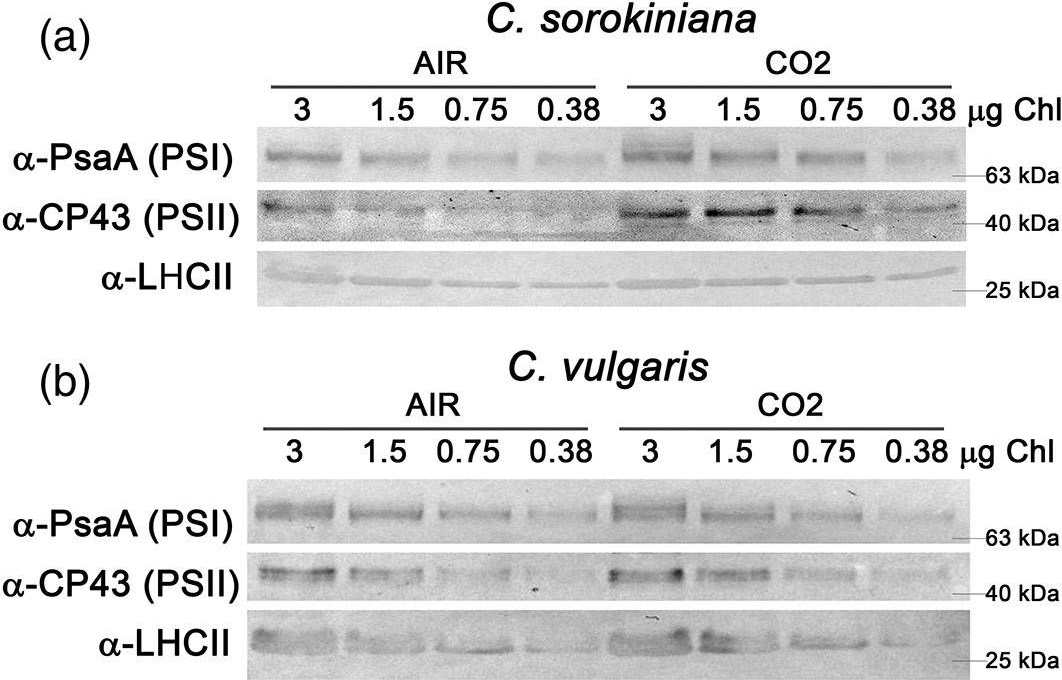
Reactant: Chlorella
Application: Western Blotting
Pudmed ID: 33931891
Journal: Plant Cell Environ
Figure Number: 5A
Published Date: 2021-09-01
First Author: Cecchin, M., Paloschi, M., et al.
Impact Factor: 6.599
Open PublicationAnalysis of PSI, PSII and LHCII content by immunoblots, P700 activity and functional PSII antenna size. (a, b) Immunoblot analysis of PSI (??PsaA antibody), PSII (??CP43 antibody) and LHCII (??LHCII antibody). Loading was performed on a chlorophyll basis: total ?g of chlorophylls loaded in each lane is reported on the top of Panel a and b. (c, d) PSI/PSII (c) and LHCII/PSII (d) ratios calculated by densitometry of immunoblot signals for C. sorokiniana (panel a, grey colour) and C. vulgaris (panel b, red colour) in AIR (full colour) or CO2 (dash colour) condition. (e) Maximal P700 oxidation on a chlorophyll basis in C. sorokiniana (left, grey colour) and C. vulgaris (right, red colour) in AIR (full colour) or CO2 (dash colour) normalized to AIR condition. (f) Functional antenna size of the photosystem II (1/?2/3) normalized to AIR condition in C. sorokiniana (grey colour) and C. vulgaris (red colour). Data are means of three biological replicates with standard deviation shown. Significant different values in CO2 versus AIR are indicated by ** (p?<?.01) and by * (p?<?.05) [Colour figure can be viewed at wileyonlinelibrary.com]
- Additional Information
-
Additional information: PsaA is a hydrophobic protein and we recommend to use PVDF membrane for transfer to assure best results.
This product can be sold containing ProClin if requested.Additional information (application): Immunogold localization has been done in leaf material of Arabidopsis thaliana.
- Background
-
Background: PsaA is a core protein of photosystem I. In plants and cyanobacteria, the primary step in oxygenic photosynthesis, the light induced charge separation, is driven bytwo large membrane intrinsic protein complexes, the photosystems I and II. Synonym: Photosystem I P700 chlorophyll a apoprotein A1.
- Product Citations
-
Selected references: Kim et al. (2024).Photoautotrophic cultivation of a Chlamydomonas reinhardtii mutant with zeaxanthin as the sole xanthophyll. Biotechnol Biofuels Bioprod. 2024 Mar 14;17(1):41. doi: 10.1186/s13068-024-02483-8.
Okegawa et al. (2023). x- and y-type thioredoxins maintain redox homeostasis on photosystem I acceptor side under fluctuating light. Plant Physiol. 2023 Nov 22;193(4):2498-2512.doi: 10.1093/plphys/kiad466.
Sulli et al. (2023). Generation and physiological characterization of genome‑edited Nicotiana benthamiana plants containing zeaxanthin as the only leaf xanthophyll. Planta . 2023 Oct 5;258(5):93. doi: 10.1007/s00425-023-04248-3.
Jiang et al. (2023). Toxic effects of lanthanum (III) on photosynthetic performance of rice seedlings: Combined chlorophyll fluorescence, chloroplast structure and thylakoid membrane protein assessment. Ecotoxicol Environ Saf. 2023 Nov 15:267:115627.doi: 10.1016/j.ecoenv.2023.115627.
Kafri et al. (2023). Systematic identification and characterization of genes in the regulation and biogenesis of photosynthetic machinery. Cell. 2023 Dec 7;186(25):5638-5655.e25.doi: 10.1016/j.cell.2023.11.007.
Nagy et al. (2023). Photoautotrophic and sustained H2 production by the pgr5 mutant of Chlamydomonas reinhardtii in simulated daily light conditions. International Journal of Hydrogen Energy Volume 53, 31 January 2024, Pages 760-769.
Hu et al. (2023). Drought affects both photosystems in Arabidopsis thaliana. New Phytol. 2023 Oct;240(2):663-675. doi: 10.1111/nph.19171. Epub 2023 Aug 2.
Ivanov et al. (2022) The decreased PG content of pgp1 inhibits PSI photochemistry and limits reaction center and light-harvesting polypeptide accumulation in response to cold acclimation. Planta 255, 36 (2022). https://doi.org/10.1007/s00425-022-03819-0
Lim et al (2022). Arabidopsis guard cell chloroplasts import cytosolic ATP for starch turnover and stomatal opening. Nat Commun. 2022 Feb 3;13(1):652. doi: 10.1038/s41467-022-28263-2. PMID: 35115512; PMCID: PMC8814037.
Lim et al (2022) Arabidopsis guard cell chloroplasts import cytosolic ATP for starch turnover and stomatal opening. Nat Commun. 2022 Feb 3;13(1):652. doi: 10.1038/s41467-022-28263-2. PMID: 35115512; PMCID: PMC8814037.
Spaniol et al. (2022) Complexome profiling on the Chlamydomonas lpa2 mutant reveals insights into PSII biogenesis and new PSII associated proteins, Journal of Experimental Botany, Volume 73, Issue 1, 5 January 2022, Pages 245–262
Rogowski et al. (2021) Light as a substrate: migration of LHCII antennas in extended Michaelis-Menten model for PSI kinetics. J Photochem Photobiol B. 2021 Dec;225:112336. doi: 10.1016/j.jphotobiol.2021.112336. Epub 2021 Oct 19. PMID: 34736069. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Oxygenic photosynthesis poster by prof. Govindjee and Dr. Shevela
Z-scheme of photosynthetic electron transport by prof. Govindjee and Dr. Björn and Dr. Shevela - Reviews:
-
Soujanya Kuntam | 2021-11-09We have used this product for many years and it has worked well with no problems in C. reinhardtiiZizhen Liang | 2019-06-27I used this antibody for many years, I really appreciate its high specificity.Veronika Z | 2014-09-29WB was performed on N. tabacum leaf in 10% ?SDS-PAGE. Dilution antibody 1:10 000. Protein was observed in area 55 kDa.Jun Liu | 2014-01-14Analysis perfomed on Arabidopsis total leaf extract, 12% SDS gel, 15 ug protein per lane, AB dilution 1:20 000, the product observed at around 60 kDa.
Accessories
AS09 461 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Plants (monocots and dicots, conifers), moss: Physcomitrella patens, Chlamydomonas reinhardtii, Triticum aestivum, cyanobacteria
compartment marker of thylakoid membrane


