1
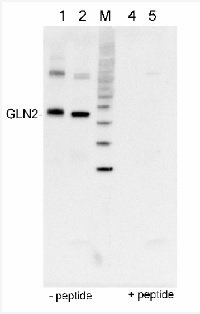
GLN2 | GS2, chloroplastic form of glutamine synthetase
AS08 296 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, N. tabacum, O. sativa, P. sativum, S. oleracea
- Product Info
-
Immunogen: KLH-conjugated synthetic peptide which is a part of part of the glutamine synthetase/guanido kinase superfamily catalytic region chosen from various available sequences, including Arabidopsis thaliana GLN2, UniProt: Q43127, TAIR: AT5G35630
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: ImmunoGold (IG), (Western blot (WB) Recommended dilution: 1 : 5000 on 0,5-5 ug protein/lane detection (WB) Expected | apparent MW: 47 | 44-45 kDa
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Nicotiana tabacum, Oryza sativa, Pisum sativum, Spinacia oleracea Predicted reactivity: Brassica napus, Diplotaxis tenuifolia,Eruca versicaria, Glycine max, Hordeum vulgare, Medicago truncatula, Pinus sylvestris, Phaseolus vulgaris, Physcomitrium patens, Populus sp., Triticum aestivum, Zea mays
Species of your interest not listed? Contact usNot reactive in: Diatoms - Application Examples
-
Application example
.jpg)
0.5 µg of protein from Arabidopsis thaliana total leaf fraction (1), 5 µg of protein fromSpinacia oleracea chlorplast enriched fraction (2), molecular weight markers (MagicMarkTM,Invitrogen) (M), the same samples as in 1 and 2 but after peptide neutralisation assay, e.g. incubation of the antibody with 100 mM excess of peptide used to elicit andt-GLN2 antibody (4,5), extracted with PEB (AS08 300), were separated on 4-12% NuPage (Invitrogen) LDS-PAGE and blotted 1h to PVDF (Millipore). Filters were blocked 1h with 2% low-fat milk powder in TBS-T (0.1% TWEEN 20) and probed with anti-GLN2 antibody (AS08 296, 1:5 000, 1h) and secondary anti-rabbit (1:20000, 1 h) antibody (HRP conjugated) in TBS-T containing 2% low fat milk powder. Antibody incubations were followed by washings in TBS-T. All steps were performed at RT with agitation. Blots were developed for 5 min with chemiluminescent detection reagent according the manufacturers instructions. Images of the blots were obtained using a CCD imager (FluorSMax, Bio-Rad) and Quantity One software (Bio-Rad).
Application examples: 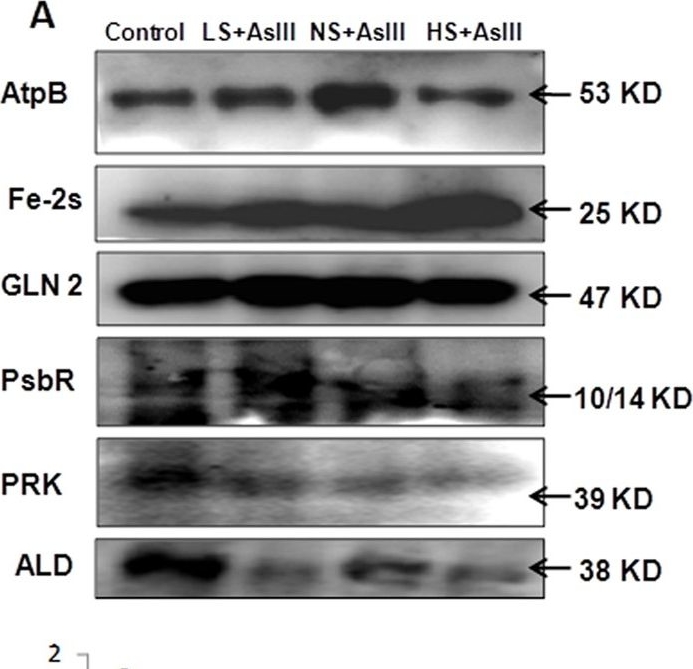
Reactant: Oryza sativa (Asian rice)
Application: Western Blotting
Pudmed ID: 26552588
Journal: Sci Rep
Figure Number: 6A
Published Date: 2015-11-10
First Author: Dixit, G., Singh, A. P., et al.
Impact Factor: 4.13
Open PublicationWestern blot analysis of selected candidate proteins (AtpB, 2Fe-2S, GLN2, Psb R, PRK, ALD) with their corresponding molecular weight in rice leaves during As stress under various S regimes (A); RuBisCo large subunit a band in coomassie blue staining (CBB) SDS gel served as a loading control (B), which used for normalization of detected proteins in densitometry studies (C) fold change indicated with respect of control samples.
Reactant: Nicotiana tabacum (Common tobacco)
Application: Western Blotting
Pudmed ID: 33963398
Journal: J Exp Bot
Figure Number: 5C
Published Date: 2021-06-22
First Author: Ancín, M., Larraya, L., et al.
Impact Factor: 6.088
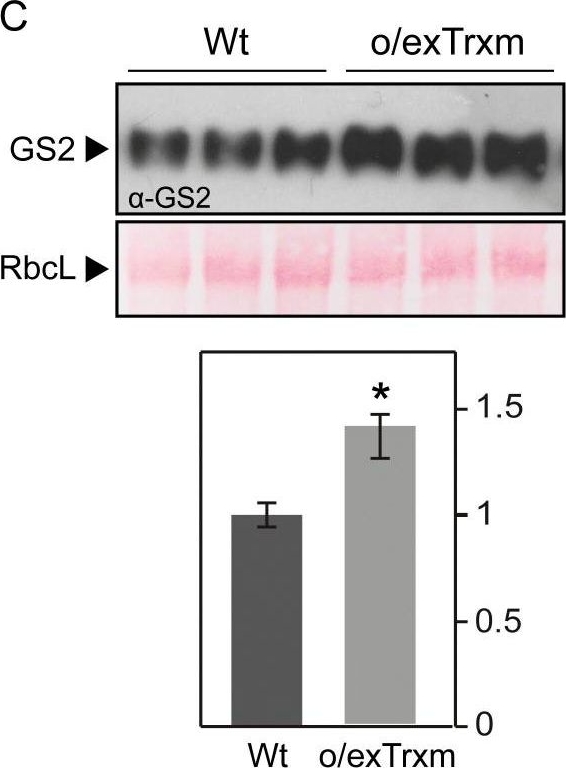
Open PublicationDetermination of GS2 oligomerization, activity, and stability under oxidative conditions. (A) GS2 protein complexes. Proteins from Wt and o/exTrxm chloroplasts were extracted under non-reducing conditions, separated by BN-PAGE, and analyzed by Western blot with anti-GS2 antibody. Photosynthetic thylakoid complexes are shown on the right and the molecular weight marker on the left. (B) Redox-sensitive GS activity. Protein extracts from Wt plants were incubated with or without 5 mM DTT or 100 µM CuCl2 for 30 min on ice, and the activity was then measured. Recovery of GS activity by reduction was performed by incubating with 5 mM DTT on ice after sample oxidation treatment (100 µM CuCl2). The activity of protein extracts from o/exTrxm plants was also measured. Results are expressed as means ±SE (n=3). (C) Quantification of GS2 in the o/exTrxm genotype. Total protein extracts (2.5 µg) were separated by SDS-PAGE and analyzed by western blot. Three different plants per line are shown in the blot. Immunoblots were analyzed with Gene Tools Analyzer software (SynGene). Data are given as means ±SE with the Wt relativized to 1. Statistical significance relative to the Wt is indicated by an asterisk (*P<0.05, Student’s t-test). (D) Degradation of GS2 protein in Wt and o/exTrxm plants. Leaves were incubated at 30 °C for up to 24 h in the presence of FeCl3, and the content of GS2 protein was analyzed by immunodetection. Similar results were obtained in eight independent biological replicates, and a representative blot is shown (upper panel). Immunoblots were analyzed with Gene Tools Analyzer software. Protein levels in plants treated for 4, 8, and 24 h are shown relative to the levels in plants treated for 0 h. Data are given as means ±SE. Statistical significance relative to Wt plants is indicated by asterisks (*P<0.05, Student’s t-test).
- Additional Information
-
Additional information: This product can be sold contacining proclin if requested
- Background
-
Background: Glutamine synthetase (GLN or GS) is one of the key enzymes involved in nitrogen metabolism of plants. It catalyses the synthesis of glutamine from glutamate and ammonia in an ATP-dependent reaction. There are two general classes of glutamine synthetase in plants: GLN1, a cytosolic form and GLN2, a chloroplastic form. GLN2 is encoded by a single gene and is highly abundant in mesophyll cells of leaves for the assimilation of ammonia produced from photorespiration and the reduction of nitrate in the chloroplasts. GLN2 is a target for thioredoxin.
- Product Citations
-
Selected references: Chang et al. (2023). Chloroplast import motor subunits FtsHi1 and FtsHi2 are located on opposite sides of the inner envelope membrane. PNAS. 2023 Sep 12;120(37):e2307747120.doi: 10.1073/pnas.2307747120. Epub 2023 Sep 5.
Hertle et al. (2021). Horizontal genome transfer by cell-to-cell travel of whole organelles. Science Advances. Volume 7. 01 Jan 2021 : eabd8215 DOI: 10.1126/sciadv.abd8215
Ancin et al. (2021) Overexpression of thioredoxin m in chloroplasts alters carbon and nitrogen partitioning in tobacco. J Exp Bot. 2021 Jun 22;72(13):4949-4964. doi: 10.1093/jxb/erab193. PMID: 33963398; PMCID: PMC8219043.
Chen et al. (2018). TIC236 links the outer and inner membrane translocons of the chloroplast. Nature. 2018 Dec;564(7734):125-129. doi: 10.1038/s41586-018-0713-y.
Tamburino et al. (2017). Chloroplast proteome response to drought stress and recovery in tomato (Solanum lycopersicum L.). BMC Plant Biol. 2017 Feb 10;17(1):40. doi: 10.1186/s12870-017-0971-0.
Dixit (2015). Sulfur alleviates arsenic toxicity by reducing its accumulation and modulating proteome, amino acids and thiol metabolism in rice leaves. Sci Rep. 2015 Nov 10;5:16205. doi: 10.1038/srep16205. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Oxygenic photosynthesis poster by prof. Govindjee and Dr. Shevela
Z-scheme of photosynthetic electron transport by prof. Govindjee and Dr. Björn and Dr. Shevela - Reviews:
-
Hsou-min Li | 2012-03-22The antibody is very specific and the titer is as good as Agrisera indicated. The availability of this antibody was critical in providing a piece of datum demanded by a reviewer. We have cited this antibody in our publication so other researchers can get the same result.
Accessories
AS08 295 | Clonality: Polyclonal | Host: Rabbit | Reactivity: [global antibody] | Arabidopsis thaliana, Eragrostis tef, Gracilaria gracilis (red algae), Gracilaria lemaneiformis, Leptodictyum riparium (Hedw.) Warnst (moss), Medicago truncatula, Physcomitrella patens, Pinus strobus, Spinacia oleracea, Solanum lycopersicum, Triticum aestivum, Zea mays
Benefits of using this antibody


