Antibodies Human Cell Biology / Carrier Proteins |
Artnr. AS99 001 |
|
Anti-KLH | Keyhole limpet hemocyanin 316 € |
Removal of cross-reactive anti-carrier (keyhole limpet haemocyanin, KLH) antibodies from rabbit serum using immobilized KLH
J.Porankiewicz-Asplund*, B.Nilsson*, A-S.Höglund** and L-G.Josefsson** *Agrisera AB, Vännäs, Sweden; **Dept of Plant Biology, Swedish University of Agricultural Sciences, Uppsala, Sweden
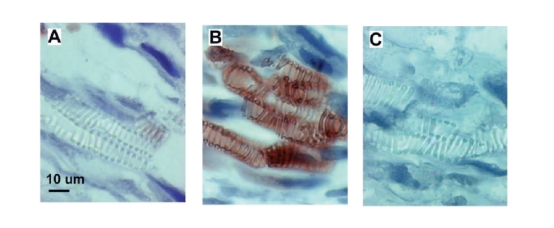
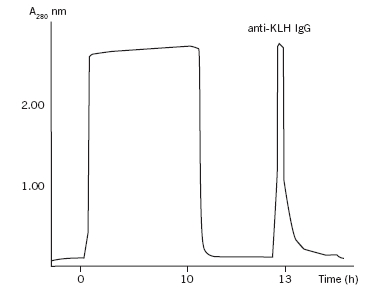
Polyclonal antibody production against peptides conjugated to keyhole limpet haemocyanin (KLH) will result in antibody production against both KLH and the peptide. Presence of anti-KLH antibodies in serum may cause non-specific background in immunohistochemical staining of tissues from some plant species. Passing serum through HiTrapTM NHS-activated column or a column with CNBr-activated SepharoseTM 4B coupled with KLH, will efficiently remove anti-KLH antibodies in a single purification step. Thus it is a flexible and convenient way of processing serum irrespective of the peptide specificities. It is also an attractive alternative to conventional purification of specific antibodies on peptide affinity columns, at least for serum where the titres of specific antibodies are high. Introduction Anti-peptide antibody production is an advantage when the antigen (protein) of interest is difficult or expensive to isolate in sufficient quantities, or when a single epitope of the protein has to be detected. Since a peptide molecule in itself is not immunogenic, it must be chemically coupled to a larger molecule (carrier) for successful immunization. Immunized animals will elicit antibody responses, both to the coupled peptide and to the carrier molecule. A common carrier protein of high molecular weight and antigenicity is keyhole limpet haemocyanin (KLH). Carrier-specific antibodies often correspond to a significant fraction of the total antibody pool (10% or more) in the serum of the immunized animal. Presence of anti-KLH antibodies in serum may cause non-specific background, for instance in immunohistochemical staining. In the plant species, Adonis aestivalis, a specific and distinct staining pattern has been observed that is the result of an immunological cross-reaction between anti-KLH antibodies and an as yet unidentified plant antigen (Fig 1). The cross-reactive antigen is most prominent in the xylem part of vascular tissues. Different parts of the plant as well as other plant species have been tested with similar results (data not shown). Therefore, the possibility of cross-reactivity must be seriously considered when planning immunohistochemical localization of plant proteins using anti-peptide serum raised with KLH as a carrier. Fig 1. Immunohistochemical staining of xylem part of vascular tissues of Adonis aestivalis. A. Tissue section stained with rabbit pre-immune serum. No staining observed. B. Tissue section stained with serum from an immunized rabbit containing antibodies against both KLH and coupled peptide. The specific staining shown is due to the anti-KLH portion of antibodies and is not characteristic for the specific anti-peptide antibodies, as evident from these and other data (not shown). C. Staining using serum (flowthrough) passed over a KLH-coupled affinity column and thus depleted of anti-KLH antibodies. The staining seen in panel B was not present. Thus, passing the rabbit immune serum through the affinity column removed in an efficient way the cross-reacting anti-KLH antibodies. Staining of plant tissues with affinity purified anti-KLH antibodies resulted in the same staining pattern observed in panel B (data not shown), which confirms the specificity of the reaction. All serum samples were diluted 1:250 in PBS, pH 7.4 before staining. However, the problem may easily be overcome by removing anti-KLH antibodies from the immune serum by passing the serum through an affinity columncoupled with KLH, for example a prepacked HiTrap NHS-activated column or a column packed with CNBractivated Sepharose 4B. As an alternative one may want to consider raising the anti-peptide antibodies using another carrier protein. Preparation of a KLH affinity column KLH (8 mg) was dissolved in the coupling buffer (200 mM sodium carbonate, 500 mM sodium chloride, pH 8.3) and applied to two HiTrap NHS-activated 1 ml columns connected in series. The columns were prepacked with NHS-activated Sepharose High Performance, which is designed for covalent coupling of ligands containing primary amino groups. Coupling of KLH protein was performed at room temperature for 1 h with a calculated efficiency of approximately 90%. After deactivation of the excess amino groups and extensive washing, the columns were ready for use. KLH was also coupled to cyanogen bromide (CNBr)-activated Sepharose 4B, following manufacturers recommendations (GE Healthcare). Removal of anti-KLH antibodies from rabbit serum in a single purification step HiiTrap columns can be used with a syringe or connected to a chromatography system. GradiFracTM system (GE Healthcare) was used for this purification. The anti-peptide serum was diluted 1:1 with PBS pH 7.4 before application on the KLH-coupled affinity column. The serum was recirculated through the column for 2 h, or overnight, to allow efficient binding of the KLH-specific antibodies to the column. The flow-through fraction was collected while specific, anti-KLH antibodies were eluted using 100 mM glycine pH 2.5, directly into 1 M Tris, pH 9.0 (Fig 2). After re-equilibration of the column with 20 ml PBS, pH 7.4 it was ready for the next run. Columns: HiTrap NHS-activated coupled with KLH, 2 x 1 ml columns in series; sample: 10 ml of serum from a rabbit immunized with a peptide-KLH conjugate dilutied 1:1 with PBS, pH 7.4; binding buffer: 10 mM sodium phosphate buffer, 150 mM NaCI, pH 7.4; elution buffer: 100 mM glycine, pH 2.5; flow rate: 1 ml/min; System: GradiFrac
Fig 2. Removal of anti-KLH antibodies from immunized rabbit serum on HiTrap NHS-activated affinity column coupled with KLH.
To confirm the removal of anti-KLH antibodies from the immunized rabbit serum, an ELISA was performed. Results indicated that the majority of the anti-KLH antibodies were removed from the serum after affinity purification (data not shown). It should be considered that in this type of affinity purification, not only specific anti-carrier antibodies will be removed, but a pool of anti-peptide antibodies that have an epitope composed of amino acids belonging to both the peptide and the carrier protein will also be removed. Questions? You are welcome to contact us [email protected] |
| « Back |