Anti-IAPP | Human IAPP (amylin) 1-37, specific for the native hormone having a disulphide-bridge between Cys2-Cys7
AS08 359 | clonality: polyclonal | host: hen | reactivity: human
- Product Info
-
Immunogen: Synthetic peptide corresponding to the human the 37 residue IAPP also known as amylin. The IAPP/amylin peptide contains a disulphide-bridge between Cys2-Cys7 Amino acid sequence: KCNTATCATQRLANFLVHSSNNFGAILSSTNVGSNTY (disulphide link between Cys2-Cys7) Host: Chicken Clonality: Polyclonal Purity: Purified,total IgY (chicken egg yolk immunoglobulin) in PBS pH 8. Contains 0.02 % sodium azide. Format: Lyophilized Quantity: 50 µl Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: ELISA (ELISA), Western blot (WB) Recommended dilution: 1:1000 (WB), 1:1000 (ELISA)
Expected | apparent MW: 3.9 kDa - Reactivity
-
Confirmed reactivity: Human Predicted reactivity: Primates, mouse, rat, dog, seal, Chinese hamster
Not reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
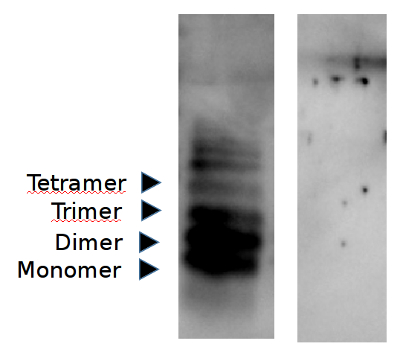
Western blot analysis illustrating specificity of AS08 359 for the oxidized form of IAPP (partly aggregated peptide illustrated below)
• Peptide separated on 16% SDS-tricine PAGE under denaturing conditions +/-β-mercaptoethanol.
• Membrane: PVDF
• Transfer buffer, tris glycine, pH 9.5 containing 0.025% SDS
• Blocking buffer: 0.4% BSA in TBS, 0.3% Tween 20
• Antibody dilution : 1:1000
• Secondary antibody: anti-chicken (HRP)
• Detection: Enhanced Chemiluminescence 10 seconds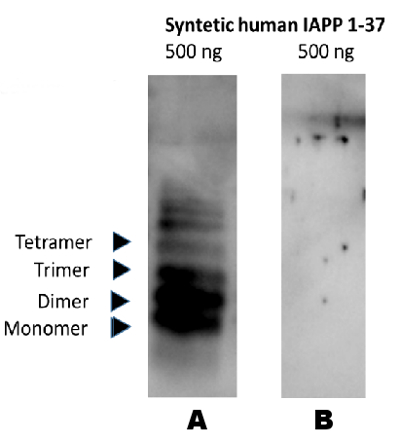
A- no reducing agent
B- reducing agent - 1 % betamercaptoethanol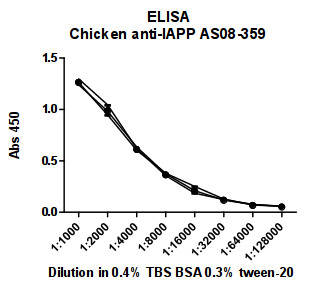
ELISA
- Nunc immunosorpt 96 well plate was coated with synthetic IAPP (amylin) 1-37, in PBS at peptide concentration corresponding to 2μg/ml.
- Blocking buffer: 0.4% BSA in TBS, 0.3% Tween 20
- Antibody dilution : 1:1000
- Washing solution TBS containing 0.3% tween-20
- Secondary antibody: anti-chicken (HRP)
- Additional Information
-
Additional information (application): Antibody is specific for the native hormone having a disulphide-bridge between Cys2-Cys7 - Background
-
Background: Amylin, or Islet Amyloid Polypeptide (IAPP) P10997, is a 37-residue peptide hormone secreted by pancreatic beta-cells at the same time as insulin (in a roughly 1:100 amylin:insulin ratio). Islet, or insulinoma, almyloid polypeptide (IAPP, or amylin) is commonly found in pancreatic islets of patients suffering diabetes mellitus type 2, or harboring an insulinoma. While the association of amylin with the development of type 2 diabetes has been known for some time, a direct causative role for amylin has been harder to establish. Recent results suggest that amylin, like the related beta-amyloid (Abeta) associated with Alzherimer's disease, can induce apoptotic cell-death in particular cultured cells, an effect that may be relevant to the deleopment of type 2 diabetes.
- Reviews:
-
This product doesn't have any reviews.
