1
Anti-UBQ11 | Ubiquitin (affinity purified)
- Product Info
-
Immunogen: Recombinant Arabidopsis thaliana ubiquitin, ubq11, with N-terminal His-tag; TAIR: At4g05050 Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water to each tube before use. Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 10 000 (WB) Expected | apparent MW: 8.5 kDa - Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Hordeum vulgare, Lupinus angustifolius, Malus domestica, Nicotiana benthamina, Oryza sativa, Pisum sativum, Solanum tuberosum, Zea mays, Vitis vinifera
Predicted reactivity: Brachypodium distachyon, Brassica napus, Capsella rubella, Cochliobolus heterostrophus, Citrus clementina, Citrus sinensis, Coffea canephora, Cymbidium faberi, Eucalyptus grandis, Erythrane guttata, Glycine max, Glycine soja, Ipomoea batatas, Medicago truncatula, Magnaporthe oryzae, Nicotiana tabacum, Phaseolus vulgaris, Populus trichocarpa, Prunus persica, Pyrus communis, Ricinus communis, Solanum nigrum, Sorghum bicolor, Triticum aestivum, Theobroma cacao, Vitis vinifera
Species of your interest not listed? Contact usNot reactive in: Algae - Application Examples
-
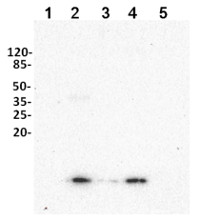
The 10 μg of soluble protein extracts from 3 -week old Solanum tuberosum (1), one -week old seedlings of Lupinus angustifolius (2), Pisum sativum (3) , Zea mays L . (4) and Hordeum vulgare (5) leaves were extracted with buffer containing 50 м M Hepes- KOH, pH 7.5, 330 м M sorbitol, 2 м M EDTA, 1 м M MgCl 2, 5 м M ascorbate, 0.05% BSA . All samples were mixed with sample buffer, denatured for 5 min at 70°C and separated on 10% Tricine -SDS -PAGE . Proteins were blotted 30 min to HybondTM -PVDF membrane (Amersham Life Science) using semi -transfer (Bio -Rad) in standard transfer buffer in presence of 20% methanol. Transfer of proteins to the membrane was checked using 1% Ponceau S staining before the blocking step. Blots were blocked in buffer (5% low -fat milk in 1xPBS, 0. 1% Tween) with for 60 min at room temperature (RT) with agitation. Blots were incubated in the primary antibody at a dilution of 1: 5 000 at RT with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in P BS -T at RT with agitation. Blot was incubated in secondary antibody
(goat anti -rabbit IgG, AS09 602, Agrisera ) diluted to 1 :50 000 for 1h at RT with agitation. The blot was washed as above and developed for 5 min with Clarity Western ECL Substrate and ChemiDoc detection system (Bio-Rad).
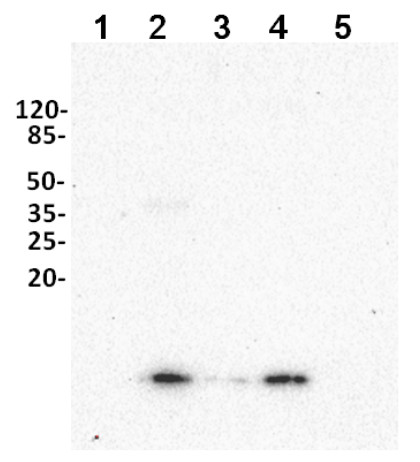
Total cell extracts of Solanum tuberosum (1), Lupinus angustifolius (2), Pisum sativum (3), Zea mays L. (4), Hordeum vulgare (5), were separated in conditions as described above. Marker - P restained SDS -PAGE ( Bio Rad, kat # 1610318). Monomers are marked by arrow. Exposure 30 seconds. For visualization of monomers, longer exposure time was applied.
Courtesy of Dr. Elena Poznidaeva Laboratory of Plant Gene Expression, Timiryazev Institute of Plant Physiology RAS, Moscow, Russia - Additional Information
-
Additional information: Ubiquitin mutant is lethal, therefore could not be used in validation of this antibody. Additional information (application): Antibody can be used to check qubiquitination status in the whole plant extracts.
Technical note: It is very difficult to detect ubiquitin monomers in total cell extracts due to a great abundance of poly and multi-ubiquitinated proteins. Recommended is size separation of protein extracts before gel electrophoresis focused on good resolution of region between 6-10 kDa.
Suggested extraction buffer: 100 mM Tris-HCl, pH 8.0, 0.1% [w/v] SDS, 0.5% [w/v] sodium deoxycholate, 1% [v/v] glycerol, 50 mM sodium metabisulfite, 20 mM N-ethylmaleimide (NEM) and protease inhibitor cocktail (Roche) (Orosa et al. 2018). This buffer will help to stabilize the conjugates and will help to detect any increase or decrease in conjugate accumulation using the antibodies. - Background
-
Background: Ubiquitin is a highly conserved regulatory protein expressed in all eukaryotic tissues. Originally this protein was called: Ubiquitous Immunopoietic Polypeptide. Its function is labeling of proteins for degradation through ubiquitin proteasome system (UPS).
- Product Citations
-
Selected references: Carrera-Castaño et al. (2026). Membrane-associated DELLA degradation modulates growth under carbon/nitrogen imbalance. Plant Cell. 2026 Jan 24:koag013. doi: 10.1093/plcell/koag013.
Shang et al. (2025). Fumarylacetoacetate hydrolase targeted by a Fusarium graminearum effector positively regulates wheat FHB resistance. Nat Commun. 2025 Jul 1;16(1):5582. doi: 10.1038/s41467-025-60736-y.
Silvestre et al. (2025). A Holistic Investigation of Arabidopsis Proteomes Altered in Chloroplast Biogenesis and Retrograde Signalling Identifies PsbO as a Key Regulator of Chloroplast Quality Control. Plant Cell Environ. 2025 Aug;48(8):6373-6396. doi: 10.1111/pce.15611.
Li et al. (2024). Stress granules sequester autophagy proteins to facilitate plant recovery from heat stress. Nat Commun . 2024 Dec 30;15(1):10910. doi: 10.1038/s41467-024-55292-w.
Xie et al. (2024). Proteasome resides in and dismantles plant heat stress granules constitutively. Mol Cell. 2024 Sep 5;84(17):3320-3335.e7. doi: 10.1016/j.molcel.2024.07.033.
Jeran et al. (2021) The PUB4 E3 Ubiquitin Ligase Is Responsible for the Variegated Phenotype Observed upon Alteration of Chloroplast Protein Homeostasis in Arabidopsis Cotyledons. Genes (Basel). 2021 Sep 6;12(9):1387. doi: 10.3390/genes12091387. PMID: 34573369; PMCID: PMC8464772.
Filippi et al. (2019). Caspase-3-like activity and proteasome degradation in grapevine suspension cell cultures undergoing silver-induced programmed cell death. J Plant Physiol. 2019 Feb;233:42-51. doi: 10.1016/j.jplph.2018.12.003.
Krasuska et al. (2016). Nitric oxide-polyamines cross-talk during dormancy release and germination of apple embryos. Nitric Oxide. 2016 Nov 23. pii: S1089-8603(16)30178-1. doi: 10.1016/j.niox.2016.11.003. [Epub ahead of print] - Protocols
-
Agrisera Western Blot protocol and video tutorials
- Reviews:
-
This product doesn't have any reviews.

