Anti-GFP | Green Fluorescence Protein (affinity purified)
AS15 2987 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Native and Recombinant GFP
- Product Info
-
Immunogen: Highly purified native GFP protein derived from Aequorea victoria, UniProt: P42212
Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Liquid Quantity: 100 µg at 1 mg/ml Storage: Store at -20°C. Avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: ELISA (ELISA), Immunofluorescence (IF), Immunoprecipitation (IP), Western blot (WB) Recommended dilution: 1 : 5000-1 : 25 000 (ELISA), 1: 100 - 1 : 500 (IF), 1 : 2000-1 : 10 000 (WB) - Reactivity
-
Confirmed reactivity: Native GFP, Recombinant GFP (E,coli), all variants of GFP, including EGFP - Application Examples
-
Immunolocalization 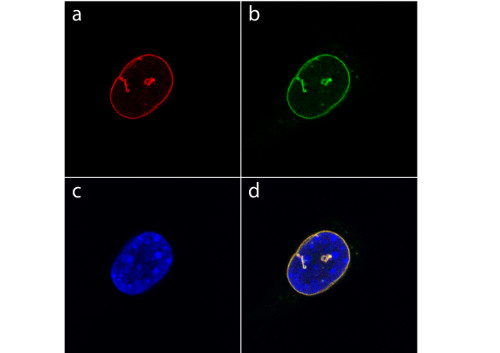
Detection of YFP-tagged Lamin A in the nucleus of mouse fibroblasts using GFP | Green Fluorescence Protein (affinity purified) antibodies. Fixation and permeabilization was performed with methanol at -20°C for 10 min. a) Indirect immunofluorescence labeling of YFP-lamin A with anti-GFP primary antibody (dilution 1: 500) as primary antibody and detected by TRITC-conjugated goat-anti-rabbit as secondary antibody.
b) Green fluorescence image of YFP-tagged Lamin A, which is incorporated into the nuclear lamina and tubular structures that penetrate into the nucleus.
c) DAPI staining of nuclear DNA.
d) Merged images
western blot
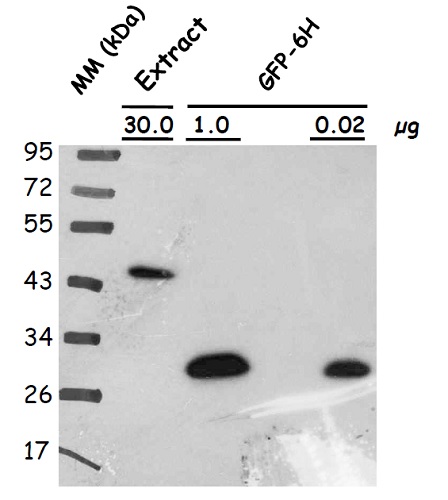
Protein extracts were obtained from Arabidopsis thaliana seeds (producing atLEA4-5 fused to GFP; other lanes contain GFP-6H loaded in indicated amounts). Following extraction buffer was used: 0.7 M sucrose, 0.5 M Tris-base, 30 mM HCl, 50 mM EDTA, 0.1 M KCl, 2% β-mercaptoethanol, 12 mg/ml poly-vinyl-poly-pyrrolidone (PVPP). This buffer was complemented with 2mL of equilbrated phenol before extraction. Samples were centrifuged and the protein phase was recovered; the extracted proteins were precipitated with 0.1 m ammonium acetate dissolved in methanol, centrifuged and the pellet washed with cold (-20 ℃) 80% acetone. The protein pellet was dissolved in SDS-solubilization buffer (1% CHES, 2% SDS, 2% ß-mercaptoethanol, 10% glicerol). Thirty 𝜇g of seed protein extract was denatured with Laemmli buffer at 95 °C for 5 min and proteins were separated on 15% SDS-PAGE and blotted 1.5 h to nitrocellulose membrane in a liquid transfer system. Blots were blocked with 2% skim milk ON at 4°C with agitation. After rinsing with TBS, blots were incubated in the primary antibody at a dilution of 1:10 000 (anti-GFP) for 3h at 4°C with agitation. The antibody solution was decanted and the blot was rinsed briefly, then washed once for 15 min. and 3 times for 5 min in TBS-T at 4°C with agitation. Then, blots were incubated in secondary antibody (anti-rabbit IgG horse-radish peroxidase conjugate) diluted to 1:25 000 in for 1h at RT with agitation. The blot was washed as above and developed for 2 min with SuperSignal West Pico Chemiluminescent Substrate, Thermo Fisher Scientific. Exposure time was for 30 seconds.
Courtesy of Dr. Alejandra Covarrubias, UNAM, Mexico - Additional Information
-
Additional information: 10 mM TRIS buffer pH 8,0 containing 0,02% sodium azide Additional information (application): Minimal cross-reactivity with E.coli proteins.
Antibody reactivity to mVenus or EBFP2 has not yet been determined. - Background
-
Background: GFP (Green fluorescent protein) was originally identified in photo organs on jellyfish Aequorea victoria. It is a naturally fluorescent protein which emits green light at a maximum wavelength of 509 nm when excited by blue or UV light. It is extensively used in laboratory as a reporter molecule to label and study cellular and subcellular proteins in living cells using a wide range of applications. Antibodies to GFP protein are used in immunoblotting and ELISA. GFP protein has molecular weight of 27 kDa.
- Product Citations
-
Selected references: Wang et al. (2025). A receptor antagonist counterbalances multiple systemin phytocytokines in tomato. Cell . 2025 Aug 12:S0092-8674(25)00866-9. doi: 10.1016/j.cell.2025.07.044.
Pinczés et al. (2024). Viral coat proteins decrease the gene silencing activity of cognate and heterologous viral suppressors. Sci Rep. 2024 Dec 28;14(1):31008. doi: 10.1038/s41598-024-81998-4.
Canal et al. (2024).Cytochrome c levels affect the TOR pathway to regulate growth and metabolism under energy-deficient conditions. New Phytol. 2024 Mar;241(5):2039-2058.
Hu et al. (2023) Spatiotemporal formation of the large vacuole regulated by the BIN2-VLG module is required for female gametophyte development in Arabidopsis [published online ahead of print, 2023 Jan 17]. Plant Cell. 2023;koad007. doi:10.1093/plcell/koad007.
Bellinvia, Garcia-Gonzalez J, Cifrova P, et al. (2022) CRISPR-Cas9 Arabidopsis mutants of genes for ARPC1 and ARPC3 subunits of ARP2/3 complex reveal differential roles of complex subunits. Sci Rep. 2022;12(1):18205. Published 2022 Oct 28. doi:10.1038/s41598-022-22982-8.
Kalachova et al. (2022) DIACYLGLYCEROL KINASE 5 Participates in Flagellin-Induced Signaling in Arabidopsis. Plant Physiol. 2022 Jul 28:kiac354. doi: 10.1093/plphys/kiac354. Epub ahead of print. PMID: 35900211.
Sun et al. (2021) The epigenetic factor FVE orchestrates cytoplasmic SGS3-DRB4-DCL4 activities to promote transgene silencing in Arabidopsis. Sci Adv. 2021 Aug 4;7(32):eabf3898. doi: 10.1126/sciadv.abf3898. PMID: 34348894; PMCID: PMC8336953.
Stelate et al. (2021) Correlative Light-Environmental Scanning Electron Microscopy of Plasma Membrane Efflux Carriers of Plant Hormone Auxin. Biomolecules. 2021 Sep 26;11(10):1407. doi: 10.3390/biom11101407. PMID: 34680040; PMCID: PMC8533460.
Wieczorek et al. (2020) Development of a New Tomato Torrado Virus-Based Vector Tagged with GFP for Monitoring Virus Movement in Plants. Viruses. 2020 Oct 20;12(10):1195. doi: 10.3390/v12101195. PMID: 33092281; PMCID: PMC7588970.
Kulich et al. (2018). Deubiquitinase OTU5 affects Root Responses to Phosphate Starvation. Plant Physiol. 2018 Jan 4. pii: pp.01693.2017. doi: 10.1104/pp.17.01693. - Reviews:
-
This product doesn't have any reviews.


