Anti-GFP | Green Fluorescence Protein (total IgY)
AS15 2998 | Clonality: Polyclonal | Host: Chicken | Reactivity: native and recombinant GFP
- Product Info
-
Immunogen: highly purified native GFP protein derived from Aequorea victoria, UniProt: P42212
Host: Chicken Clonality: Polyclonal Purity: Total IgY in 10 mM TRIS, 0,15 mM NaCl, pH 8. Contains 0.02 % sodium azide. Format: Liquid Quantity: 100 µl (1 mg/ml) Storage: Store at 4°C; make aliquots to avoid working with a stock. Or Store in small aliquots at -20°C, Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: ELISA (ELISA), Western blot (WB) Recommended dilution: 1 : 5000-1 : 25 000 (ELISA), 1 : 2000-1 : 10 000 (WB) - Reactivity
-
Confirmed reactivity: Native GFP, Recombinant GFP (E,coli), all variants of GFP - Application Examples
-
Application examples 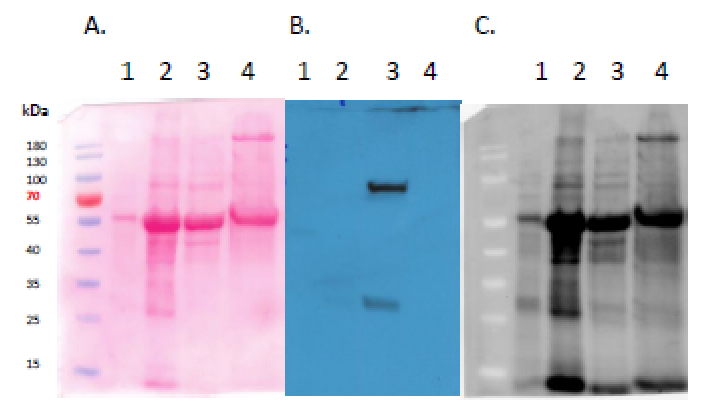
Samples
20 ug of Arabidopsis thaliana Columbia-0 overexpressing GRP8-GFP leaf protein extract (1), 70 ug of Arabidopsis thaliana Columbia-0 overexpressing GFP-SnRK2.4 leaf protein extract (2), 50 ug of Nicotiana benthamiana transiently expressing GFP-SnRK2.4 leaf protein extract (3), 50 ug of Nicotiana benthamiana transiently expressing SnRK2.4-GFP leaf protein extract (4). Mark: MW markers: PageRuler Prestain Protein Ladder from Thermo Fisher Scientific (#26616) The total protein extracts were freshly prepared from A. thaliana leaves with extraction buffer 1 (samples 1&2) or N. behtamiana leaves with extraction buffer 2 (samples 3&4). The extraction buffer 1 contained: 20 mM Tris-HCl pH 7.5; 2 mM EDTA; 2 mM EGTA; 50 mM β-glycerophosphate; 250 mM sucrose; 10 mM Na3VO4; 1% Triton X-100; 150 mM NaCl; 10 mM DTT; 1 mM PMSF and 1 x Complete Protease Inhibitor Cocktail (Roche). The extraction buffer 2 contained: 100 mM HEPES, pH7.5; 5 mM EDTA; 5 mM EGTA; 10 mM DTT; 1 mM Na3VO4; 10 mM NaF; 50 mM b-glycerophosphate; 10 mM pyridoxal 5-phosphate; 10% glycerol and 1 x Complete protease inhibitors (EDTA-free, Roche). The extraction buffers were added to the powdered material in a 1:1 v:v ratio. Samples were incubated on a rotator for 30 min at 4°C and then centrifuged for 30 min, 12 000 rpm at 4°C. Supernatant was transferred into new tubes and protein concentration was measured using Bradford Protein Assay. Samples were then denatured with 3x Laemmli sample buffer for 5 min in 95°C. Next samples were separated on 10 % SDS-PAGE stain-free gels (Bio-Rad) and blotted overnight to PVDF membrane using wet transfer. Blot was blocked with 3% milk and 0,1% Tween 20 in TBS for 1h/RT with agitation. Blot was incubated in the primary antibody at a dilution of 1: 5 000 for 1,5h/RT with agitation in 3% milk and 0,1% Tween 20 in TBS solution. The antibody solution was decanted and the blot was rinsed briefly, then washed 5 times for 5 min in TBS-T at RT with agitation. Blot was incubated in Agrisera matching secondary antibody (rabbit anti-chicken IgG horse radish peroxidase conjugated, AS10 1489, Agrisera) diluted to 1:10 000 in for 1h/RT with agitation. The blot was washed as above and developed for 1 min with Pierce ECL Western Blotting Substrate. Exposure time was 5 min (short exposure)/ 10 min (long exposure). Additionaly blot was briefly rinsed with water and developed for 1 min with chemiluminescent detection reagent. Exposure time was 1 min.
Courtesy of Katarzyna Szymańska, Institute of Biochemistry and Biophysics PAS > Plant Protein Phosphorylation Laboratory, Warsaw, Poland - Additional Information
-
Additional information (application): Minimal cross-reactivity with E.coli proteins
- Background
-
Background: GFP (Green fluorescent protein) was originally identified in photo organs on jellyfish Aequorea victoria. It is a naturally fluorescent protein which emits green light at a maximum wavelength of 509 nm when excited by blue or UV light. It is extensively used in laboratory as a reporter molecule to label and study cellular and subcellular proteins in living cells using a wide range of applications. Antibodies to GFP protein are used in immunoblotting and ELISA. GFP protein has molecular weight of 27 kDa.
- Protocols
- Antibody protocols
- Reviews:
-
This product doesn't have any reviews.