1
Anti-HSP23,6 | Heat shock protein 23,6 (mitochondrial)
AS15 2980 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana
- Product Info
-
Immunogen: KLH-conjugated synthetic peptide derived from Arabidopsis thaliana HSP23.6, mitochondrial, UniProt: Q96331, TAIR: AT4G25200
Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 1000 (WB) Expected | apparent MW: 23.6 | 18 kDa - Reactivity
-
Confirmed reactivity: Arabidopsis thaliana Predicted reactivity: Arabidopsis thaliana
Not reactive in: Brassica rapa - Application Examples
-
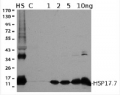
Application example 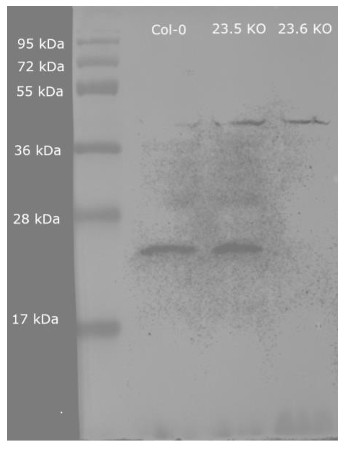
50 µg of total protein from Col-0, HSP23.5 KO and HSP23.6 KO extracted with sample buffer (60 mM Tris-HCl pH 6.8, 2% SDS, 65 mM DTT, 15% sucrose, 0.01% bromophenol blue) and denatured with heating at 95°C for 5 minutes was separated on 15% SDS-PAGE and blotted 2 hours to nitrocellulose membrane using semi-dry transfer. Blot was blocked with 5% (w/v) milk in TBS-T (20 mM Tris-HCl pH 7.5, 150 mM NaCl, 0.05% Tween-20) for 1h/RT with agitation. Blot was rinsed with TBS-T. Blot was then incubated in the primary antibody diluted to 1:2000 in TBS-T for 1 hour at RT with agitation. The antibody solution was decanted, and the blot was rinsed briefly twice, then washed 3 times for 10 minutes in TBS-T at RT with agitation. Blot was incubated in GE Healthcare secondary antibody (anti-rabbit IgG horseradish peroxidase conjugated) diluted to 1:5000 in TBS-T for 1 hour at RT with agitation. Blot was washed as above and incubated with chemiluminescent detection reagent in extreme low femtogram range, visualizing with G:Box iChemi XT(Syngene).
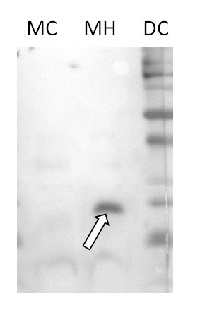
Courtesy of Parth Patel/Vierling lab. University of Massachusetts, USA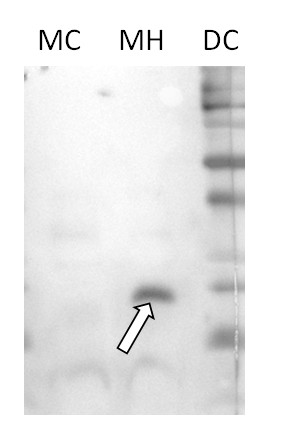
Mitochondrial enriched fractions were prepared from 4-week old Arabidopsis thaliana Col-0 rosette leaves (control, or heat shocked 3h 38°C) using the method of Huang et al (2013, Nature Comm. 4:2558). Crude mitochondria obtained from 3-4 g leaves were suspended in 50 µL washing buffer (0.4 M sucrose, 50 mM Tris pH 7.5, 3mM EDTA, 0.1% BSA). For western blot analysis, 40 µL of suspension were mixed with 10 µL of 5X SDS-PAGE sample buffer (40 mM Tris pH 6.8, 0.8 % SDS, 0.4 % bromophenol blue, DTT 0.2 M, 20 % glycerol), heat denatured at 95 °C for 3 min, separated on 13.5 % SDS-PAGE, and blotted 1h to PVDF (0.2 µm Immobilon PSQ transfer membrane, Millipore) using tank transfer (10 mM CAPS pH 11, 10 % ethanol). After staining with Ponceau red, blot was blocked with TBS containing 1.5 % Tween 20 by incubation at room temperature (RT). Blot was briefly rinsed three times with TBS-T (TBS with 0.05 % Tween 20) and incubated overnight at 4°C with the primary antibody at a dilution of 1:1 000 in TBS-T. The antibody solution was decanted and the blot was rinsed briefly twice, then washed 3 times for 15 min in TBS-T at RT with agitation. Blot was incubated in secondary antibody (goat anti-rabbit IgG horse radish peroxidase conjugated, from Agrisera) diluted to 1:75 000 in TBS-T for 1h at RT with agitation. The blot was washed as above and developed for 2 min with chemiluminescent detection reagent. DC: dual color markers (Biorad), 5 µL MC: 10 µL of mitochondria enriched fractions from control leaves MH: 10 µL of mitochondria enriched fractions from heat-shocked leaves.
Remark: Protein were not measured in the extracts because of the presence of BSA. Coomassie blue staining confirmed similar protein content of crude mitochondria extract (estimated to around 2µg/µL). Similar protein content of MC and MH extracts is also supported by the background with the AB HSP23.5 antibody.
Courtesty Dr. David Macherel, Mitostress team, IRHS, France. - Additional Information
-
Additional information: Detection on total extracts needs to be optimized.
Antibody is recognizing HSP23.6 synthesized in vitro using the PURExpress in Vitro Protein Synthesis Kt (NEB).
Antibody is not cross reacting with cytosolic HSPs.Additional information (application): Please note that there might be no HSPs accumulation below temperature of 32-34°C. HSPs are induced when the plant experience temperatures higher than the growing temperature with around 10°C. So, the HSPs induction temperatures for plants grown at 18C differ from these for plants grown at 24°C.
Another very effective parameter is the humidity. When using low humidity the plant has a chance to cool down through transpiration. In this case the HSPs induction requires higher temperatures.
- Background
-
Background: HSP23.6 (heat shock protein 23.6 (mitochondrial)) belongs to small heat shock protein family and is localized in mitochondria. - Product Citations
-
Selected references: Cha et al. (2020). Humic acid enhances heat stress tolerance via transcriptional activation of Heat-Shock Proteins in Arabidopsis. Sci Rep. 2020 Sep 14;10(1):15042.doi: 10.1038/s41598-020-71701-8. - Protocols
- Reviews:
-
This product doesn't have any reviews.