1
Anti-PGDH3 | Phosphoglycerate dehydrogenase 3 (chloroplastic)
AS20 4391 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana
- Product Info
-
Immunogen: Recombinant, full-length PGDH3 of Arabidopsis thaliana, overexpressed in E.coli with terminal His-tag UniProt: Q9LT69, TAIR: At3g19480 Host: Rabbit
Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 50 µl, of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 3000 (WB) Expected | apparent MW: 62.1 | 55-60 kDa - Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Nicotiana benthamiana Predicted reactivity: Acer yangbiense, Actinidia chinensis, Arachis hypogaea, Brassica campestris, Brassica napus, Brassica oleracea, Cajanus cajan, Capsella rubella, Cucumis melo, Cucumis sativus, Daucus carota, Eutrema salsugineum, Fagus sylvatica, Glycine max, Gossypium hirsutum, Lupinus angustifolius, Nicotiana tabacum, Noccaea caerulescens, Malus domestica, Mucuna pruriens, Nyssa sinensis, Ricinus communis, Theobroma cacao, Trema orientale, Vigna unguiculata Not reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
Application example
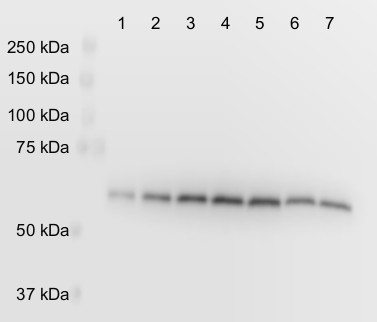
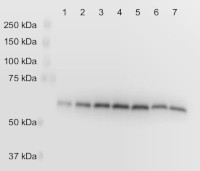
Lane: 1: 0.5 mg tissue wild type 2: 1 mg tissue wild type 3: 1.5 mg tissue wild type 4: 2 mg tissue wild type 5: 2.5 mg tissue wild type 6: 2.5 mg tissue pgdh3-1 (PGDH3 knockout) 7: 2.5 mg tissue pgdh3-2 (PGDH3 knockout) Remaining signal in lanes 6 and 7 is from the other two isoforms of the enzyme that were not knocked out. Arabidopsis thaliana tissue was frozen with liquid nitrogen and ground to a fine powder with mortar and pestle. Protein was extracted by mixing with extraction buffer (200 mM tris pH 8.0, 4% sodium dodecyl sulfate) to 0.5 grams fresh weight/mL and heating at 90°C for 10 min. Insoluble debris was removed by centrifuging at 21,000 x g for 1 minute. The supernatant was removed and mixed with equal volume 2x SDS-PAGE sample buffer. Samples were loaded on an 8% acrylamide gel. 20 mA were applied through the gel until the dye front ran off the bottom of the gel. Contents of the gel were electroblotted onto nitrocellulose (0.2 µm pore size) with 70 V for 45 minutes using a Biorad tank (wet) transfer system. The blot was blocked for 10 minutes at room temperature in tris buffered saline with 0.5% tween (TBST) plus 5% fat free powdered milk (blocking buffer). The blot was incubated with the anti-AtPGDH3 antibodies diluted in blocking buffer at 1:3000 overnight at 4°C while gently rocking at 75 rpm. The blot was rinsed with TBST 3 times for 5 minutes each. The blot was incubated with HRP conjugated goat anti-rabbit secondary antibody (from Proteintech) diluted 1:25000 in TBST for 2 h at room temperature while gently rocking at 75 rpm. The blot was rinsed 3 times for 20 minutes each. The blots were developed with chemiluminescent detection reagent for 5 minutes. The signal was collected using a Li-Cor C-DiGit Blot Scanner using the standard sensitivity setting.
Remaining signal in lanes 6 - 8 is from the other two isoforms of the enzyme that were not knocked out.
Courtesy of Dr. Philip Day, Kunz Lab, Washington State University, USA
- Additional Information
-
Additional information (application): Beside PGDH3 the antibody is recognizing in Arabidopsis thaliana PGDH1, UniProt: O49485-1 and PGDH2, UniProt: O04130-2
- Background
-
Background: PGDH3 | Phosphoglycerate dehydrogenase 3 (chloroplastic) is an enzyme ((EC:1.1.1.95) involved in the plastidial phosphorylated pathway of serine biosynthesis (PPSB), step 1 of the subpathway that synthesizes L-serine from 3-phospho-D-glycerate. Expressed in aerial parts. Not detected in roots and meristematic tissue. Expressed in cotyledons, adult leaves, stigma and anther filaments. - Product Citations
-
Selected references: Höhner et al. (2021) Stromal NADH supplied by PHOSPHOGLYCERATE DEHYDROGENASE3 is crucial for photosynthetic performance. Plant Physiology. 2021. kiaa117, https://doi.org/10.1093/plphys/kiaa117 - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Agrisera Educational Poster Collection - Reviews:
-
This product doesn't have any reviews.
Related products
AS03 037 | Clonality: Polyclonal | Host: Rabbit | Reactivity: global antibody and compartment marker for higher plants, lichens, algae, cyanobacteria, dinoflagellates, diatoms
Benefits of using this antibody
AS05 084 | Clonality: Polyclonal | Host: Rabbit | Reactivity: [global antibody] for higher plants, algae, liverwort, cyanobacteria, diatoms | cellular [compartment marker] of thylakoid membrane
Benefits of using this antibody

