1

Anti-PrxQ | Peroxiredoxin, thioredoxin reductase
AS05 093 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, M. esculenta, S. oleracea, Z. mays
- Product Info
-
Immunogen: His-tagged full length protein (with presequence) of Arabidopsis thaliana was overexpressed in in E.coli. Isolated with HiTrap column (GE Healthcare) Q9LU86, At3g26060
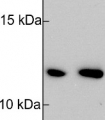
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 200 µl Reconstitution: For reconstitution add 200 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 5000 (WB) Expected | apparent MW: 16 kDa
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Manihot esculenta, Spinacia oleracea, Zea mays
Predicted reactivity: Marchantia polymorpha, Populus sp. , Triticum aestivum, Oryza sativa
Species of your interest not listed? Contact usNot reactive in: Malus domestica, Medicago truncatula, Solanum lycopersicum - Application Examples
-
Application examples: 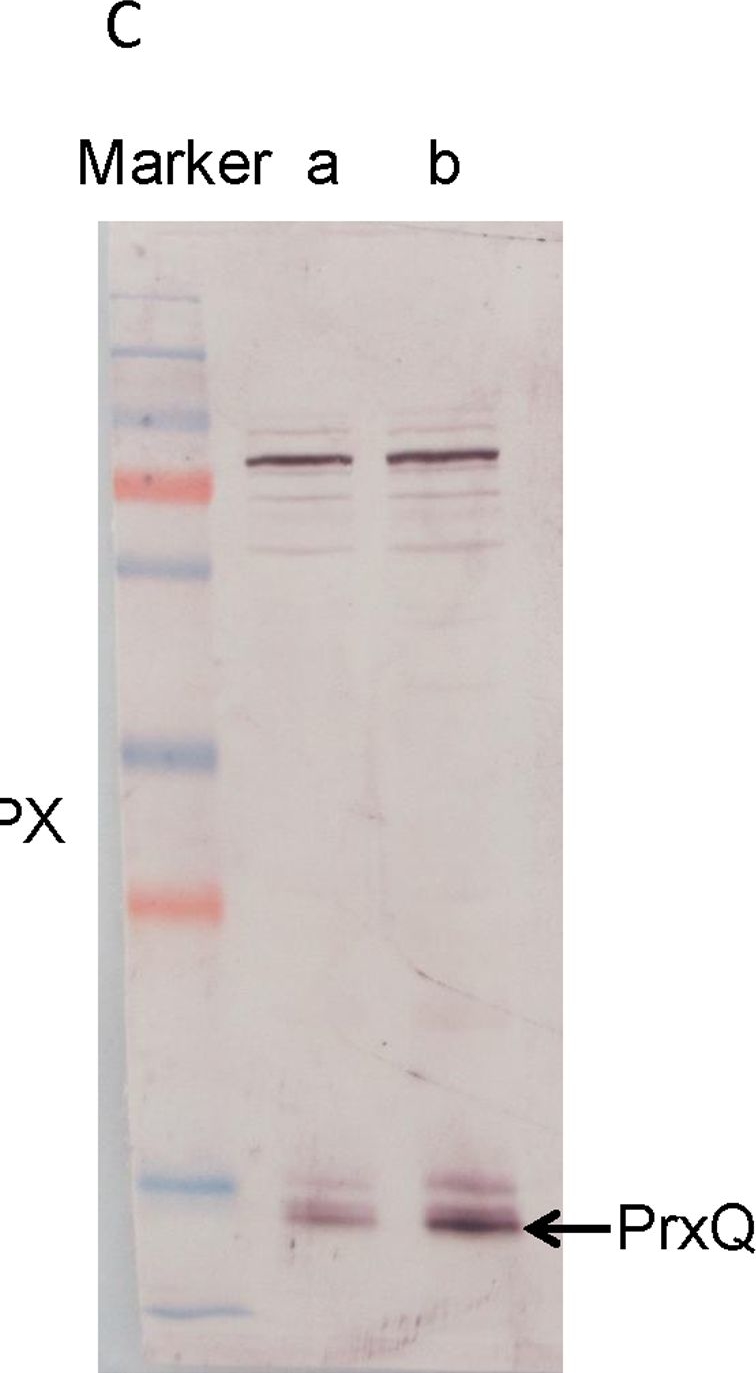
Reactant: Manihot esculenta (Cassava)
Application: Western Blotting
Pudmed ID: 24727655
Journal: PLoS One
Figure Number: 5C
Published Date: 2014-04-15
First Author: An, F., Fan, J., et al.
Impact Factor: 2.942
Open PublicationWestern blotting of Rubisco, APX and PrxQ.The expression of Rubisco, APX and PrxQ in leaves of cassava NZ199 diploid (a) and autotetraploid (b) genotypes were detected by western blotting using antiRubisco-polyclonal antibody (AS07218), anti-APX antibody (AS08368) and anti-PrxQ antibody (AS05093) from Agrisera, respectively.
Reactant: Manihot esculenta (Cassava)
Application: Western Blotting
Pudmed ID: 27881899
Journal: Plant Mol Biol Report
Figure Number: 8B
Published Date: 2016-11-25
First Author: An, F., Li, G., et al.
Impact Factor: 1.526
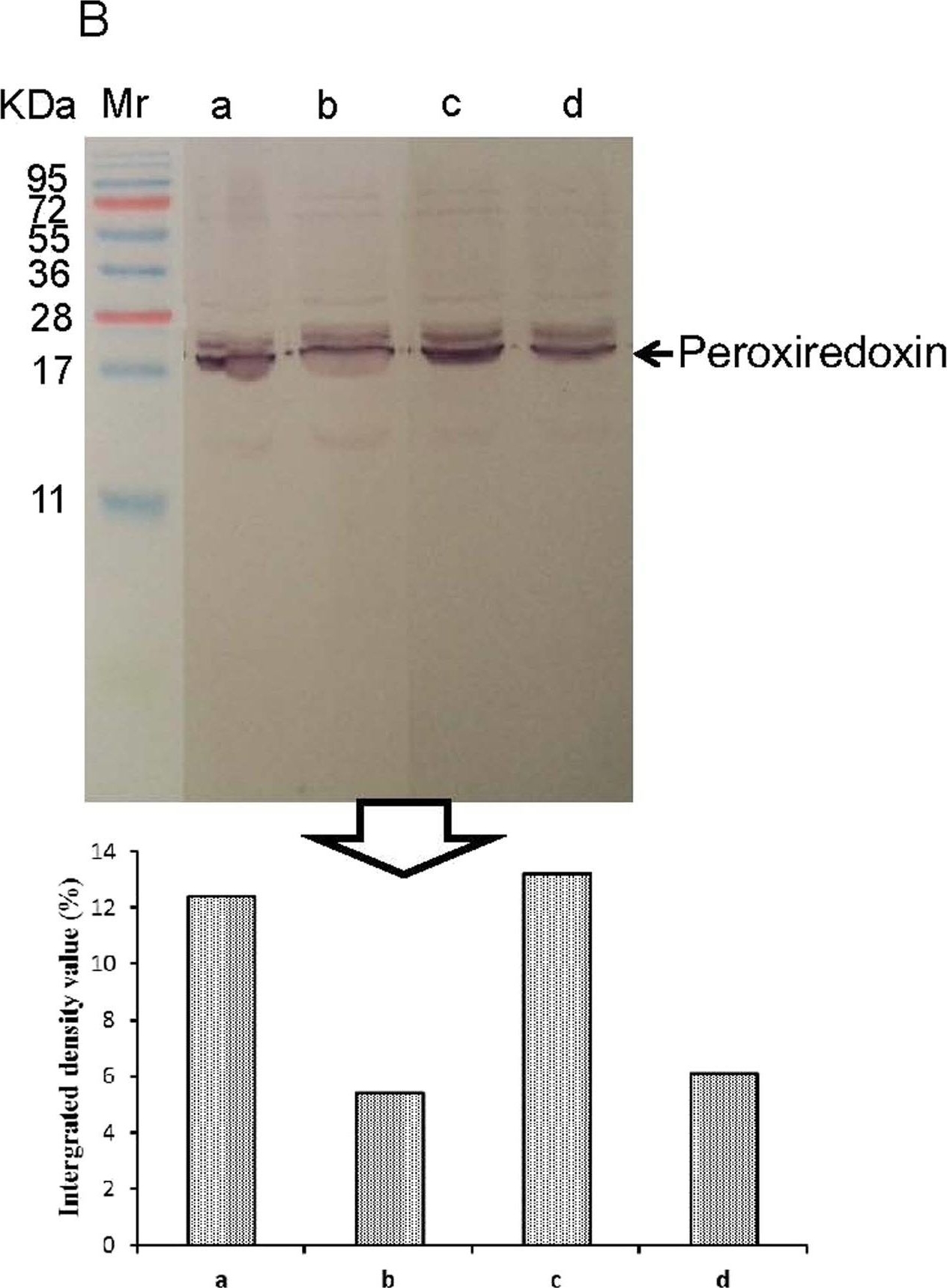
Open PublicationWestern blotting of Rubisco and peroxiredoxin. The expressions of Rubisco (a) and peroxiredoxin (b) in apical expanded leaves of SC8and Col1046 were detected by Western blotting using anti-Rubisco polyclonal antibody (AS07218) and anti-peroxiredoxin antibody (AS05093) from Agrisera, respectively. Mr, protein marker; lines a and b, the expression of Rubisco and peroxiredoxin of SC8 in 5 °C for 0 and 10 days, respectively; lines c and d, the expression of Rubisco and peroxiredoxin of Co11046 in 5 °C for 0 and 10 days, respectively
- Additional Information
-
Additional information: This product can be sold containing proclin if requested Additional information (application): In stroma fractions a weak background reaction at 28 kDa is visible, No crossreactivity in any thylakoid fractions - Background
-
Background: Peroxiredoxins (EC=1.11.1.15) belong to the enzyme family which is ubiquitous in all kingdoms of life. Prx Q enzyme acting by reducing hydroperoxides. Peroxiredoxins have no heme group, unlike the other peroxidases, but perform their enzymatic activity using cysteine residues with redox-active thiol groups. The ability of peroxiredoxins to hydrolyze hydroperoxides suggests that this protein family has a general function in oxidant defence.
- Product Citations
-
Selected references: Okegawa et al. (2023). x- and y-type thioredoxins maintain redox homeostasis on photosystem I acceptor side under fluctuating light. Plant Physiol. 2023 Nov 22;193(4):2498-2512.doi: 10.1093/plphys/kiad466.
Yoshida et al. (2018). Thioredoxin-like2/2-Cys peroxiredoxin redox cascade supports oxidative thiol modulation in chloroplasts. Proc Natl Acad Sci U S A. 2018 Aug 13. pii: 201808284. doi: 10.1073/pnas.1808284115.
Yoshida et al. (2016). Hisabori T1.Two distinct redox cascades cooperatively regulate chloroplast functions and sustain plant viability. Proc Natl Acad Sci U S A. 2016 Jul 5;113(27):E3967-76. doi: 10.1073/pnas.1604101113. Epub 2016 Jun 22.
Yoshida et al. (2015). Thioredoxin Selectivity for Thiol-Based Redox Regulation of Target Proteins in Chloroplasts. J Biol Chem. 2015 Apr 15. pii: jbc.M115.647545.
Feifei et al. (2014). Comparison of Leaf Proteomes of Cassava (Manihot esculenta Crantz) Cultivar NZ199 Diploid and Autotetraploid Genotypes. PLoS One. 2014 Apr 11;9(4):e85991. doi: 10.1371/journal.pone.0085991. eCollection 2014.
Wu et al. (2013). Proteomic and Phytohormone Analysis of the Response of Maize (Zea mays L.) Seedlings to Sugarcane Mosaic Virus. PLoS One. July 23;8(7). - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts - Reviews:
-
Monika Ostaszewska | 2016-06-10I used this antibody on A. thaliana leaf extracts and it worked perfectly (Ostaszewska-Bugajska et al. 2015 JPP 186:25-38). SDS-PAGE was performed on 10% gels and 20 µg of proteins were loaded per lane. Primary antibody dilution was 1:5 000 and secondary (suitable for ECL detection) 1:20 000.
Related products
AS03 037 | Clonality: Polyclonal | Host: Rabbit | Reactivity: global antibody and compartment marker for higher plants, lichens, algae, cyanobacteria, dinoflagellates, diatoms
Benefits of using this antibody