1
Anti-LEA4-25 | Group 4-late embryogenesis abundant protein
- Product Info
-
Immunogen: Recombinant Phaseolus vulgaris PvLEA 4-1 sequence derived from Pv01G142000
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 2000 (WB) Expected | apparent MW: 16 | 18 kDa
- Reactivity
-
Confirmed reactivity: Phaseolus vulgaris
Predicted reactivity: Glycine tomentosa
Species of your interest not listed? Contact usNot reactive in: Arabidopsis thaliana
- Application Examples
-
application information
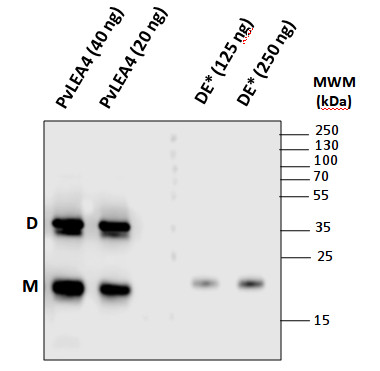
Phaseolus vulgaris seed protein extracts which is an extract enriched in hyrdophilic proteins, obtained in a following way: Dry seeds were used to separate embryos from cotyledons. Approximately 500 mg of embryos were ground in a mortar in the presence of liquid nitrogen or dry ice. The powder was resuspended and homogenized in 1 mL of 20 mM TES pH 8.0, 0.5 M NaCl, 10 mM Na2VO3, 10 mM NaF, 1 mM PMSF and one tablet/10 mL (protease inhibitor cocktail tablet, Roche). The suspension was centrifuged at 14,000 rpm in a microcentrifuge during 10 min at 4ºC, supernatant was separated and centrifugation was repeated once more. Then, supernatant was boiled 10 min and transfer to ice for 15 min, the suspension was centrifuged at 14,000 rpm in a microcentrifuge during 10 min at 4ºC, and the boil-ice-centrifugation procedure was repeated twice. Subsequently, the supernatant was dialyzed against 10 mM phosphate buffer pH 8.0 in the cold room. Concentration of the protein extract was determined by standard procedures and its integrity was verified by SDS-PAGE.
Following samples were separated: PvLEA4 is a recombinant PvLEA4-1 proein which does not contain any tag and was purified from bacterial extract. M: monomoer, D: dimer. DE*: total protein extract from dry Phaseolus vulgaris embryos. Proteins were separated on 12% SDS-PAGE and blotted 1h to nitrocellulose membrane using semi-dry transfer for 1 h. Blots were blocked ON at 4ºC in 2% non-fat milk with agitation. Blots were incubated in the primary antibody at a dilution of 1: 1 000 for 1h at RT with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in TBS-T at RT with agitation. Blot was incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated, from Agrisera, AS09 602) diluted to 1:25 000 in 2% non-fat milk for 1h at RT with agitation. The blot was washed as above and developed for 5 min with ECL according to the manufacturer's instructions. Exposure time was 1 min.
Courtesy of Dr. Alejandra A. Covarrubias, UNAM, Mexico - Additional Information
-
Additional information: This antibody does not recognize group 4 LEA proteins from Arabidopsis thaliana.
Additional information (application): The PvLEA4-1 protein from common bean presents a deduced molecular mass of 16 kDa; however, as other LEA and disordered proteins it migrates with an apparent higher molecular mass, possibly due to post-translational modifications - Background
-
Background: LEA4-25 (Group 4-late embryogenesis abundant protein PvLEA4-25) belongs to a group of very hydrophilic proteins, described over 25 years ago as accumulating during late stages of plant seed development. Found in vegetative plant tissues following exposure to environmental stress.
Synonymes: Putative late embryogenesis abundant protein LEA. - Protocols
-
- Reviews:
-
This product doesn't have any reviews.
Related products
AS19 4257 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana, Pisum sativum, Nicotiana tabacum
TROL protein can be used as a chloroplast dual-localization marker: for the thylakoids (the major portion of the protein) and the chloroplastic inner envelope. Since the envelopes make a very small portion of the total membranes, the envelope form of TROL is detectable only when isolated envelopes are applied.


