1
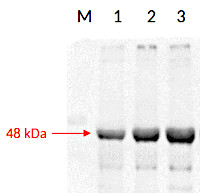
Anti-RPN6 | 26S proteasome non-ATPase regulatory subunit 9 (A,thaliana)
AS15 2845 | Clonality: polyclonal | Host: rabbit | Reactivity: Arabidopsis thaliana, Nicotiana tabacum
- Product Info
-
Immunogen: KLH-conjugated synthetic peptide derived from Arabidopsis thaliana RPN6 sequence, UniProt: Q9LP45-1, TAIR: AT1G29150 Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1:1000 (WB)
Expected | apparent MW: 46.7 kDa - Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Nicotiana benthamiana
Predicted reactivity: Populus tremula - Application Examples
-
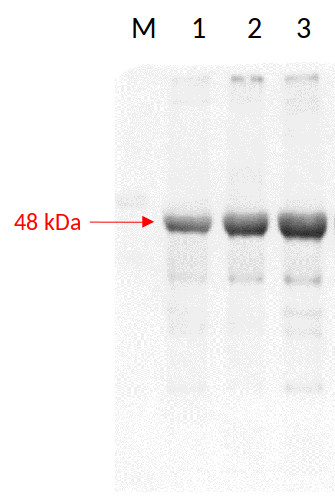
Samples:
1 - 14 µg of Nicotiana tabacum, L. whole leaf extract of adult control plant (untreated)
2 - 14 µg of Nicotiana tabacum, L. whole leaf extract of adult plant after treatment with 100 µM AgNP-PVP
3 - 14 µg of Nicotiana tabacum, L. whole leaf extract of adult plant after treatment with 100 µM AgNP-CTAB14 µg/well of total protein extracted freshly from 0.05 g of lyophilized whole leaf of adult tobacco plants following the Phenol extraction protocol with extraction buffer containing Trizma base (500 mM), Ethylenediaminetetraacetic acid (EDTA) (50 mM), sucrose (700 mM) and Potassium chloride (KCl) (100 Mm) with addition of phenylmethylsulfonyl fluoride (PMSF) (1mM) and 2% β-mercaptoethanol. After short incubation on 4°C with agitation, phenol was added. The phenol (supernatant) phase containing proteins, was collected after centrifugation and equal volume of extraction buffer was added. After centrifugation, supernatant phase was collected and 4 volumes of 0.1 M ammonium acetate (with 10% methanol) was added, and proteins were precipitated ON/-20°C. The next day, protein pellets were washed 3 times in ammonium acetate with rounds of centrifugations inbetween, and finally in aceton. Protein pellet was lastly resuspended in Isoelectric focusing buffer (IEF) containing 9 M urea, 4% CHAPS, 20 mM DTT, 1.2% Ampholytes pH 3 to 10. Protein concentrations was measured with modified Bradford method and denatured with Laemmli sample buffer at 95°C for 5 min. Total proteins were separated on 12% SDS-PAGE and blotted 1h to nitrocellulose (pore size of 0.2 µm), using wet transfer. Blot was blocked with 3% milk in TBS-T, 1h/RT. Blot was incubated in the primary antibody at a dilution of 1: 1 000 for 1h/RT with agitation in a solution of 3% milk in TBS-T and then ON/4°C. The antibody solution was decanted and the blot was then washed twice for 10 min in TBS-T at RT with agitation. Blot was incubated in Agrisera matching secondary antibody (goat anti-Rabbit IgG1 HRP conjugated AS09 602) diluted to 1:10 000 in TBS-T for 1h/RT with agitation. The blot was washed 2 times for 10 min in TBS-T and developed for 5 min with chemiluminescent detection reagent. Exposure time was 12 min
Courtesty of MSc, Karla Košpić, University of Zagreb, Faculty of Science Department of Biology, Croatia
- Background
-
Background: Proteasome-dependent degradation serves an essential role in the removal of a wide variety of key nuclear and cytosolic proteins. Substrates are targeted for proteolysis by the ubiquitin pathway before being degraded by the 26 S proteasome. RPN6 associates with an ATPase subunit of the 19S proteasome regulatory complex, AtS6A.
Alternative names: 19S proteosome subunit 9, 26S proteasome non-ATPase regulatory subunit 11 homolog, AtRPN6. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Agrisera Educational Poster Collection - Reviews:
-
This product doesn't have any reviews.


