1
Anti-Sec21p | Gamma subunit, COP vesicles
AS08 327 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana, Zea mays | Cellular [compartment marker] of Golgi in immunolocalization and of COP1 in western blot
- Product Info
-
Immunogen: GST fusion of a part of recombinant Sec21 of Arabidopsis thaliana Q0WW26, At4g34450
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Immunofluorescence (IF), Western blot (WB) Recommended dilution: 1 : 1000 (IF), 1 : 1000 (WB) Expected | apparent MW: 98 kDa
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Phaeodactylum tricornutum, Zea mays
Predicted reactivity: Brachypodium distachyon, Brassica napus, Brassica rapa subsp. pekinensis, Capsella rubella, Citrus clementina, Coffea canephora, Eutrema salsugineum, Glycine max, Glycine soja, Hordeum vulgare var. distichum, Medicago truncatula, Oryza sativa, Populus trichocarpa, Prunus persica, Ricinus communis, Solanum lycopersicum, Solanum tuberosum, Sorghum bicolor, Theobroma cacao, Triticum aestivum, Vitis vinifera, Zea mays,
Species of your interest not listed? Contact usNot reactive in: Nicotiana tabacum, Microsporidia sp. - Application Examples
-
Application example
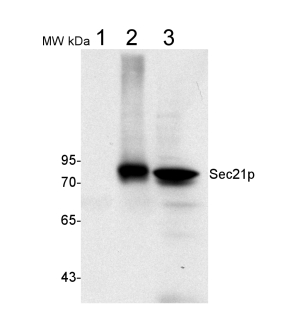
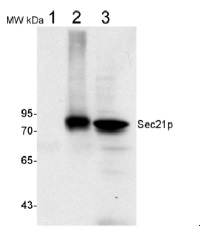
50 µg of total protein from (1) Nicotiana tabacum protoplast total protein, (2) Arabidopsis thaliana protoplast soluble protein, (3) Arabidopsis thaliana protoplast total protein were separated on 10 % SDS-PAGE and blotted 2h to nitrocellulose (Semi-dry, 200mA). Filters were blocked over night with 5% low-fat milk powder in TBS and probed with anti-Sec21p antibodies (AS08 327, 1:1000, 1h) and secondary anti-rabbit (1:20000, 1 h) antibody (HRP) in TBS-Tween. Signal was detected with chemiluminescent detection reagent, exposure time was 1 minute.
Protoplasts were extracted in 50mM Tris, 10 mM EDTA and Triton X100, 0.02%.
Immunofluorescence
.jpg)
Immunofluorescence labelling of rabbit anti-SEC21 (gamma subunit of COP vesicles; red) in 5-day-old root epidermal cells of Arabidopsis thaliana expressing ER-mGFP5-HDEL (ER marker; green). The antibody was diluted 1:1000 and the secondary antibody, donkey anti-rabbit CY5-coupled (Jackson ImmunoResearch) was diluted 1:300. The nuclei were stained with DAPI (blue).
Courtesy of Dr. Anna Gustavsson and Dr. Markus Grebe, Umeå Plant Science Centre, Sweden
Application examples: 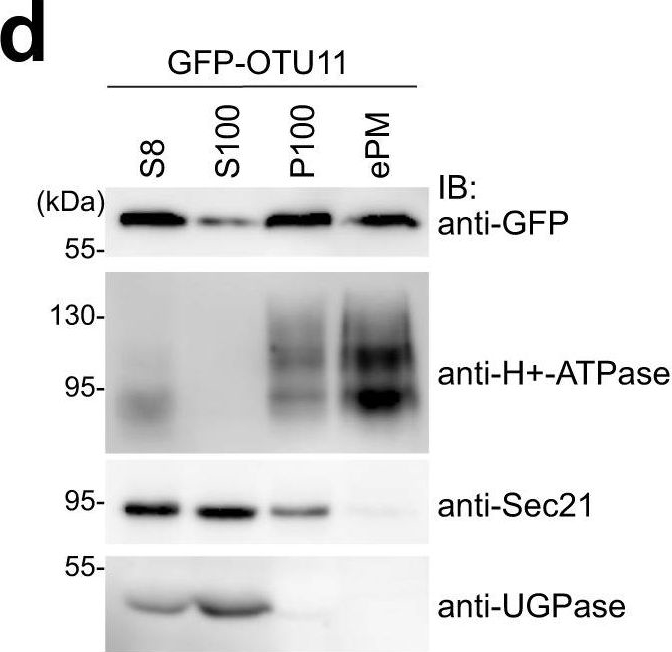
Reactant: Arabidopsis thaliana (Thale cress)
Application: Western Blotting
Pudmed ID: 36371501
Journal: Nat Commun
Figure Number: 1D
Published Date: 2022-11-12
First Author: Vogel, K.
Impact Factor: 15.405
Open PublicationGFP-OTU11 and GFP-OTU12 localize to the PM in Arabidopsis root cells.a Representative confocal images of 35S promoter-driven YFP-fusions of UBP3, UBP18, UBP22, UBP23, UBP25, UBP27, OTU9, OTU10, OTU11, and OTU12. YFP-fusion proteins were transiently expressed in Arabidopsis root cell-derived protoplasts. For each construct, protoplast transformation was performed at least twice. Scale bars: 10 µm. b Representative confocal images of protoplasts expressing 35pro:GFP-OTU11 or 35pro:GFP-OTU12 together with 35Spro:RFP-SYP121. The signal intensity profile along the dotted line (merged image) is shown on the right. Scale bars: 10 µm. Protoplast transformations were performed at least twice. c Representative confocal images of wild-type (Col-0) Arabidopsis seedlings harboring OTU11pro:GFP-OTU11 or OTU12pro:OTU12-GFP. Both lines were co-stained with FM4-64 for 5 min before imaging. The confocal analysis was performed at least twice. The signal intensity profile along the dotted line (merged image) is shown on the right of the panels. Scale bars: OTU11 10 µm, OTU12 20 µm. d GFP-OTU11 is present in the enriched PM fraction. Total proteins were prepared from wild-type (Col-0) seedlings containing 35Spro:GFP-OTU11. The extracts were centrifuged at 8000 ×g, and the supernatant (S8) was further centrifuged at 100,000 ×g to yield supernatant (S100) and microsome (P100) fractions. P100 samples were subsequently treated with Brij-58 for the enriched PM fraction (ePM). H+-ATPase, UGPase, and Sec21 were used as markers for the PM, soluble fraction, and microsomal fractions, respectively. Three independent experiments were carried out, and a representative result is shown. Source data are provided as a Source Data file. e, f35Spro:GFP-OTU11- and 35Spro:GFP-OTU12-expressing wild-type (Col-0) seedlings were stained with FM4-64 and treated with 50 µM brefeldin A (e) or 33 µM Wortmannin (f). The experiments were repeated at least three times; one representative image is shown. Scale bars: 10 µm. g35Spro:GFP-OTU11-, 35Spro:GFP-OTU12-expressing wild-type (Col-0) seedlings, and the PI(4)P-sensor (P5R)-containing seedlings were treated with 60 µM phenylarsine oxide (PAO). Whereas PAO treatment causes dissociation of P5R from the PM, localization of GFP-OTU11 and GFP-OTU12 remains unaffected. The experiment was repeated at least three times; one representative image is shown. Scale bars: 10 µm.
Reactant: Arabidopsis thaliana (Thale cress)
Application: Immunohistochemistry
Pudmed ID: 32358503
Journal: Nat Commun
Figure Number: 3A
Published Date: 2020-05-01
First Author: Hurný, A.
Impact Factor: 15.405
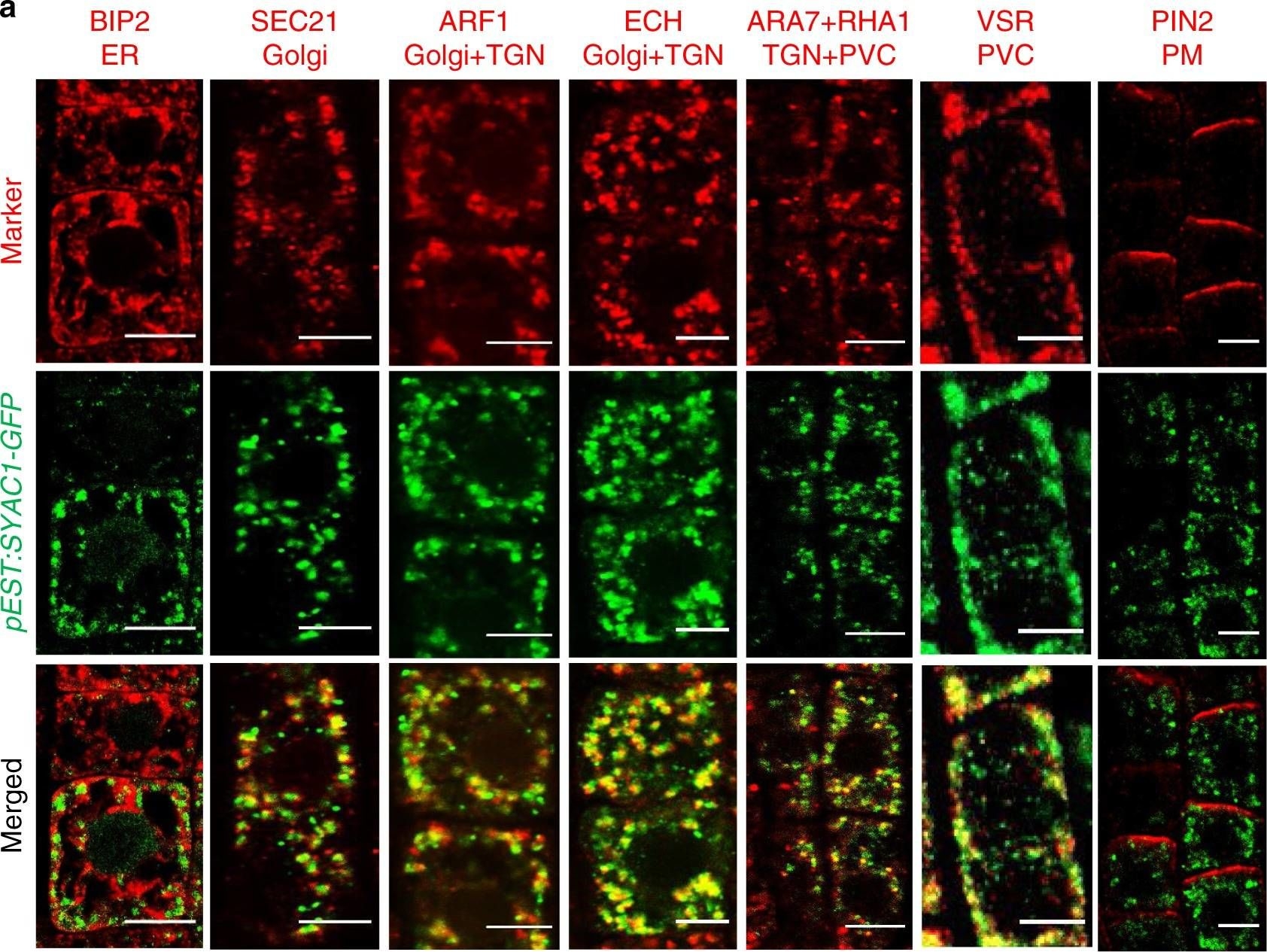
Open PublicationSYAC1 colocalizes with Golgi/TGN/endosomal/PVC markers.Co-imunolocalization of SYAC1-GFP with markers for different subcellular compartments (a) and quantification of colocalization using Pearson correlation coefficient (b). Five-day-old pEST:SYAC1-GFP seedlings grown on mock medium treated with 5 µM estradiol for 6 h used in co-imunolocalization experiments (n = 10 roots with 1–5 epidermal cells each). Co-imunolocalization performed using antibodies against anti-GFP and subcellular markers. In the boxplots, center lines show the medians; box limits indicate the 25th and 75th percentiles as determined by Origin software; whiskers extend 1.5 times the interquartile range from the 25th and 75th percentiles, individual data points are represented by dots. ER Endoplasmic reticulum, TGN trans-Golgi network, PVC prevacuolar compartment, PM plasma membrane. Scale bar 5 µm.
- Additional Information
-
Additional information: This product can be sold containing proclin if requested Additional information (application): This antibody can be used as a Golgi marker in immunolocalization and as a marker of COP1 in Western blot.
References describing immunolocalization (IF) and (IG) studies:
Pimpl et al (2000). In Situ Localization and in Vitro Induction of Plant COPI-Coated Vesicles. Plant Cell. 2000 Nov;12(11):2219-36.
Ritzenthaler et al. (2002). Reevaluation of the Effects of Brefeldin A on Plant Cells Using Tobacco Bright Yellow 2 Cells Expressing Golgi-Targeted Green Fluorescent Protein and COPI Antisera. Plant Cell. 2002 Jan;14(1):237-61. - Background
-
Background: Sec21p is a constituent of the COPI vesicle coatomer.
- Product Citations
-
Selected references: Gallei et al. (2025). Super-resolution expansion microscopy in plant roots. Plant Cell. 2;37(4).
Tangpranomkorn et al. (2025). A land plant-specific VPS13 mediates polarized vesicle trafficking in germinating pollen. New Phytol. 2025 Feb;245(3):1072-1089.
Zhao et al. (2024). Subcellular spatial regulation of immunity-induced phosphorylation of RIN4 links PAMP-triggered immunity to Exo70B1. Front Plant Sci. 15:1473944.
Vogel et al. (2022) Lipid-mediated activation of plasma membrane-localized deubiquitylating enzymes modulate endosomal trafficking. Nat Commun. 2022;13(1):6897. Published 2022 Nov 12. doi:10.1038/s41467-022-34637-5.
Brumm, Singh, Kriechbaum, et al. (2022) N-terminal domain of ARF-GEF GNOM prevents heterodimerization with functionally divergent GNL1 in Arabidopsis. Plant J. 2022;112(3):772-785. doi:10.1111/tpj.15980.
Skalicky et al. (2021) Auxin Metabolite Profiling in Isolated and Intact Plant Nuclei. Int J Mol Sci. 2021 Nov 16;22(22):12369. doi: 10.3390/ijms222212369. PMID: 34830250; PMCID: PMC8620152.
Hurny et al. (2020). SYNERGISTIC ON AUXIN AND CYTOKININ 1 Positively Regulates Growth and Attenuates Soil Pathogen Resistance. Nat Commun. 2020 May 1;11(1):2170. doi: 10.1038/s41467-020-15895-5. (immunolocalization)
Lupette et al. (2019). The architecture of lipid droplets in the diatom Phaeodactylum tricornutum. Algal Research Volume 38, March 2019, 101415.
Singh et al. (2018). A single class of ARF GTPase activated by several pathway-specific ARF-GEFs regulates essential membrane traffic in Arabidopsis. PLoS Genet. 2018 Nov 15;14(11):e1007795. doi: 10.1371/journal.pgen.1007795.
Kitakura et al. (2017). BEN3/BIG2 ARF GEF is Involved in Brefeldin A-Sensitive Trafficking at the trans-Golgi Network/Early Endosome in Arabidopsis thaliana. Plant Cell Physiol. 2017 Oct 1;58(10):1801-1811. doi: 10.1093/pcp/pcx118.
Nagel et al. (2017). Arabidopsis SH3P2 is an ubiquitin-binding protein that functions together with ESCRT-I and the deubiquitylating enzyme AMSH3. Proc Natl Acad Sci U S A. 2017 Aug 7. pii: 201710866. doi: 10.1073/pnas.1710866114.
Wang et al. (2016). Comprehensive proteomic analysis of developing protein bodies in maize (Zea mays) endosperm provides novel insights into its biogenesis. J Exp Bot. 2016 Dec;67(22):6323-6335. Epub 2016 Oct 27.
Wattelet-Boyer et al. (2016). Enrichment of hydroxylated C24- and C26-acyl- chain sphingolipids mediates PIN2 apical sorting at trans-Golgi network subdomains. Nat Commun. 2016 Sep 29;7:12788. doi: 10.1038/ncomms12788.
Derbyshire et al. (2015). Proteomic Analysis of Microtubule Interacting Proteins over the Course of Xylem Tracheary Element Formation in Arabidopsis. Plant Cell. 2015 Oct 2. pii: tpc.15.00314.
Tanaka et al. (2013). Cell Polarity and Patterning by PIN Trafficking through Early Endosomal Compartments in Arabidopsis thaliana. PLoS Genet. May;9(5). (immunolocalization).
Hopff et al. (2013). The plasma membrane proteome of maize roots grown under low and high iron conditions. J Proteomics Jan 24.
Pimpl et al (2000). In situ localization and in vitro induction of plant COPI-coated vesicles. Plant Cell. 2000 Nov;12(11):2219-36. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Oxygenic photosynthesis poster by prof. Govindjee and Dr. Shevela
Z-scheme of photosynthetic electron transport by prof. Govindjee and Dr. Björn and Dr. Shevela - Reviews:
-
This product doesn't have any reviews.
Related products
AS08 325 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, C.reinhardtii, Elaeis sp. , L. longiflorum, M. truncatula, N. tabacum, O. sativa, P. hybrida cv Mitchell, P. patens, S. tuberosum | Cellular [compartment marker] of Golgi in immunolocalization and COP1 in Western blot


