1
AgriseraECL SuperBright (100 ml)
This product can be purchased in 3 different volumes:
AS16 ECL-S-10, 10 ml. Trial size limited to one per customer
AS16 ECL-S-100, 100 ml
Choose the appropriate volume in the drop down menu to the right
- Data sheet
-
AgriseraECL SuperBright for Western Blot detection is a high quality substrate for detection of horseradish peroxidase enzyme activity at extreme low femtogram levels, offering low background and superior signal to noise ratio.
It is a ready to use 2 component system with low background and superior signal to noise ratios and highest sensitivity.
This reagent offers extreme low femto level sensitivity combined with low background and superior signal to noise ratios. - Product Info
-
Quantity: 2 x 5 ml, two component ready to use solutions, enough for 5 midi blots (6,8 x 8,1 cm) 2 x 50 ml, two component ready to use solutions, enough for 50 midi blots (6.8 x 8.1 cm), which is 2754 cm2 Storage:
Shelf life is 24 months when stored in the dark at 2 °C to 8 °C. Keep container tightly closed. Store away from heat or light. - Application Examples
-
Application example
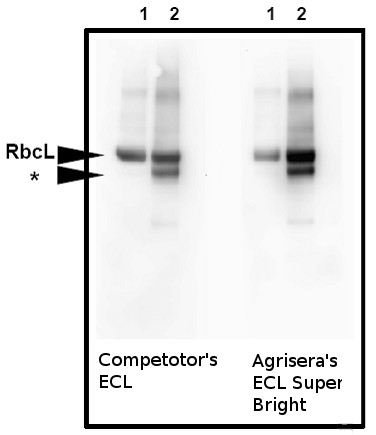
10 µg of total protein from Arabidopsis thaliana leaf (1), Hordeum vulgare leaf (2) were extracted with Protein Extraction Buffer PEB (AS08 300). Samples were diluted with 1X sample buffer (NuPAGE LDS sample buffer (Invitrogen) supplemented with 50 mM DTT and heat at 70°C for 5 min and keept on ice before loading. Protein samples were separated on 4-12% Bolt Plus gels, LDS-PAGE and blotted for 70 minutes to PVDF using tank transfer. Blots were blocked immediately following transfer in 5% non-fat milk dissolved in 20 mM Tris, 137 mM sodium chloride pH 7.6 with 0.1% (v/v) Tween-20 (TBS-T) for 1h at room temperature with agitation. Blots were incubated in Agrisera anti-RbcL primary antibody (AS03 037) at a dilution of 1: 20 000.
The antibody solution was decanted and the blot was rinsed briefly twice, and then washed 1x15 min and 3x5 min with TBS-T at room temperature with agitation. Blots were incubated in secondary antibody (goat anti-rabbit IgG horse radish peroxidase conjugated, recommended secondary antibody AS09 602, Agrisera) diluted to 1:20 000 following by washes and detection using Agrisera ECL SuperBright detection reagent. Images of the blots were obtained using a CCD imager (VersaDoc MP 4000) and Quantity One software (Bio-Rad). Exposure time was 60 seconds.
Note: In the barley sample, a well characterised 44 kDa degradation product is observed (Kokobun et al. 2002).
Caution:
Do not add any protein or HRP enzyme to reagent solution.
Optional: wash your tube, microtiter plate or membrane with 0.2 M solution of Sodium Phosphate, Dibasic, in Deionized Water. (Initial pH may be approximately 9). Bring the pH down to 8.4 with the slow addition of a 0.2M solution of Sodium Phosphate, Monobasic, in water. - Additional Information
-
Additional information (application): User Instruction
- Store reagents A and B in the darkness at 4-8°C.
- Mix equal volumes of reagent A and B (chemiluminescent substrate) in a clean container and equilibrate to room temperature 30 minutes before use.
- Prepare your membrane prior addition of chemiluminescent substrate, by a wash with a buffer used in your protocol (PBS or TBS or TBST-T). This will allow to remove any background prior to substrate contact.
- Optimal visualization may be obtained up to 20 minutes after substrate contact. Usually, incubation for 2-5 is optimal.
- Remove excess substrate by filter paper.
- Cover blot with clear plastic wrap or sheet protector and expose either with x-ray film or CCD camera.
For best results clean containers and high quality water has to be used.
HS code for this product is: 3822.00.0002. - Product Citations
-
Selected references: Wieczorek et al. (2020) Development of a New Tomato Torrado Virus-Based Vector Tagged with GFP for Monitoring Virus Movement in Plants. Viruses. 2020 Oct 20;12(10):1195. doi: 10.3390/v12101195. PMID: 33092281; PMCID: PMC7588970.
Fallah et al. (2018). Plasminogen activation is required for the development of radiation-induced dermatitis. Cell Death Dis. 2018 Oct 15;9(11):1051. doi: 10.1038/s41419-018-1106-8. Naranjo et al. (2021) NTRC Effects on Non-Photochemical Quenching Depends on PGR5. Antioxidants (Basel). 2021 Jun 3;10(6):900. doi: 10.3390/antiox10060900. PMID: 34204867; PMCID: PMC8229092.
Ferrero et al. (2019). Class I TCP transcription factors target the gibberellin biosynthesis gene GA20ox1 and the growth promoting genes HBI1 and PRE6 during thermomorphogenic growth in Arabidopsis. Plant Cell Physiol. 2019 Jul 11. pii: pcz137. doi: 10.1093/pcp/pcz137. - Reviews:
-
Rima Sulniute | 2018-11-19So far, this one is the best from all I have used before-sensitive and almost no background.
Accessories

This product can be purchased in 3 different volumes:
AS16 ECL-N-10, 10 ml. Trial size limited to one per customer
AS16 ECL-N-100, 100 ml
Choose the appropriate volume in the drop down menu to the right

For an extra 33 EUR on your order you will receive:
- Goat anti-rabbit secondary antibody of high titer, min. 1: 25 000 1h/RT, HRP conjugated
- Two Agrisera chemiluminescence reagents: with low picogram (Agrisera Bright)
and extreme low femtogram detection range (Agrisera SuperBright)

