1
Anti-ChlG (G4) | Chlorophyll synthase (chloroplastic)
AS14 2793 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana, Hordeum vulgare
- Product Info
-
Immunogen: KLH-conjugated peptide derived from Arabidopsis thaliana chlorophyll synthase, UniProt: Q38833, TAIR: AT3G51820
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Clear-native PAGE (CN-PAGE), Western blot (WB) Recommended dilution: 1: 10 000 (CN-PAGE), 1 : 1000 (WB) Expected | apparent MW: 41.8 kDa (further processed into a mature form) - Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Hordeum vulgare Predicted reactivity: Avena sativa, Camelia sinensis, Capsicum annuum, Cocomyxa subellipsoidea, Gossypium hirsutum, Micromonas pusilla, Nicotiana tabacum, Oryza sativa, Populus trichocarpa, Ricinus communis, Spinacia oleracea, Triticum urartu, Cyanobacteria
Species of your interest not listed? Contact us - Application Examples
-
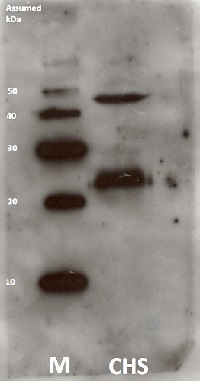
Samples:
1 mg of Arabidopsis thaliana mutant whole leaf extract (kindly provided by Dr. Charng (Academia Sinica) (1)
1 mg of Arabidopsis thaliana whole leaf extract (wild ecotype, Col-0) (2)
The mutant contains a mutation that cause G217R substitution. For more detail information please see the following reference: Lin, Y.-P., Lee, T., Tanaka, A. & Charng, Y. Analysis of an Arabidopsis heat-sensitive mutant reveals that chlorophyll synthase is involved in reutilization of chlorophyllide during chlorophyll turnover. The Plant journal : for cell and molecular biology 80, 14–26 (2014).
Proteins separated by SDS-PAGE were transferred to a PVDF membrane of 0.45 µm (Immobilon®-P Transfer membrane) through a tank system with constant voltage (40 V) for 2 h, in Transfer buffer (25 mM Tris; 192 mM glycine; 10 % EtOH). Membranes were blocked with 5 % skim milk in PBS-Tween (0.01 % Tween® 20 in PBS (137mM NaCl, 8.1mM Na2PO4, 2.68mM KCl, 1.47mM KH2PO4, pH 7.4)) for 16 h at 4°C without agitation and washed with PBS-Tween for five minutes three times with gentle shaking. After blocking and washing, membranes were incubated with the primary antibody diluted in CanGet Signal solution 1 for 1h at RT. The concentrations of primary antibody were 1:15000. Next, 3 incubations of 5 minutes with PBS-Tween were performed to wash out the excess of antibody. The incubation with the secondary antibody (Agrisera AS09 602) which diluted 1: 25000 in CanGet Signal solution 2 was performed for 1h at RT. After the incubation, the excess of secondary antibody was washed out as the primary antibody. Developing was performed by the Enhanced chemiluminescence method (ECL). This method is based on the chemiluminiscence reaction of luminol. The enzyme HRP, which is linked to secondary antibodies, catalyz-expres the oxidation of luminol when there is hydrogen peroxide in alkaline conditions, generating a product that emits luminescence (Whitehead et al., 1979). Membranes were incubated in Agrisera ECL Bright before exposure. Developing was performed using the Chemiluminescent Imaging System LuminoGraph (ATTO) with variable exposure times.Courtesy of Ms. J. Kishimoto, Institute of Low Temperature Science Hokkaido University, Japan
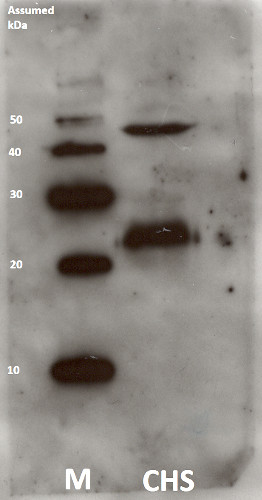
Barley seeds were grown for 4.5 days in the dark before they were harvested and intact plastids were isolated in light according to Eichacker et al. 1996. The concentration of plastids was determined by counting in a Thoma counting chamber. The membrane complexes were isolated according to Reisinger et al. 2008 before 3x SB buffer (2% (w/v) SDS, 10% (w/v) sucrose, 0.03% (w/v) bromphenol blue, 66 mM NaCO3, 66 mM dithiothreitol) was added. The proteins were denatured for 2 min at 72 °C. The membrane fractions were loaded onto a 12.5 % SDS-acrylamide gel, giving approximately 2.5x107 plastids per lane alongside with Magic Marc (LC5602, Life technologies). The gel was then run at 175 V with 1xSDS running buffer. The proteins were then transferred onto a nitrocellulose membrane by using semi-wet transfer and Towbin buffer w/10 % methanol. The proteins were transferred at 2 mA/cm2 for 1 hour. The membrane was blocked with TBS-milk, washed 3x5 min in TBS-tween and the primary antibody (Chlorophyll synthase, AS14 2793 from Agrisera) was added at a concentration of 1:1000. The membrane was incubated ON at 4 °C (approximately 18 h) before washing with TBS-tween and incubation for 1h at RT with the secondary antibody diluted 1:12 500 (Agrisera AS09 602). After washing, the membrane was incubated in an ECL1/ECL2 solution (100 mM Tris, 2.5 mM Luminol, 0.4 mM pCoumaracid/100 mM Tris, 0.0183 % Hydrogenperoxide) before exposure to a film for 7 min. The film was then developed and fixed using chemicals from Kodak.
Courtesy of Eli Drange Vee, Staff Engineer, Centre for Organelle Research, University of Stavanger, Norway - Background
-
Background: G4 (Chlorophyll synthase) is an enzyme which performs estrification of chlorophyllide (a and b), which is the last step of chlorophyll biosynthesis. Alternative names: polyprenyl transferase, protein G4.
- Product Citations
-
Selected references: Maeda et al. (2022). Characterization of photosystem II assembly complexes containing ONE-HELIX PROTEIN1 in Arabidopsis thaliana. J Plant Res. 2022 Mar;135(2):361-376. doi: 10.1007/s10265-022-01376-x. Epub 2022 Feb 10. PMID: 35146632.(CN-PAGE)
Maeda et al. (2022). Characterization of photosystem II assembly complexes containing ONE-HELIX PROTEIN1 in Arabidopsis thaliana. J Plant Res. 2022 Mar;135(2):361-376. doi: 10.1007/s10265-022-01376-x. Epub 2022 Feb 10. PMID: 35146632.(CN-PAGE)
Maeda et al. (2022). Characterization of photosystem II assembly complexes containing ONE-HELIX PROTEIN1 in Arabidopsis thaliana. J Plant Res. 2022 Mar;135(2):361-376. doi: 10.1007/s10265-022-01376-x. Epub 2022 Feb 10. PMID: 35146632.(CN-PAGE) - Reviews:
-
This product doesn't have any reviews.
Related products

AS05 067 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana, Cyanobacteria, Horderum vulgare, Nicotiana tabacum, Oryza sativa, Phalenopsis Sogo Yukidian cultivar V3, Pinus yunnanensis, Pisum sativum, Triticum aestivum


