1
Anti-GPA1 | Guanine nucleotide-binding protein subunit alpha 1
AS16 3939 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana
- Data sheet
- Product Info
-
Immunogen: Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1: 2000 (WB) Expected | apparent MW: 49.3 kDa - Reactivity
-
Confirmed reactivity: Arabidopsis thaliana Predicted reactivity: Amborella trichopoda, Ananas comosus, Arachis sp., Beta vulgaris, Brassica sp., Cajanus cajan, Camelina sativa, Capsella rubella, Capsicum annuum, Cicer arietinum, Coffea canephora, Cucumis melo, Daucus carota, Elaeis guineensis, Eucalyptus grandis, Eutrema sp., Fragaria vesca, Genlisea aurea, Glycine max, Glycine soja, Jatropha curcas, Malus domestica, Manihot esculenta, Medicago truncatula, Morus notabilis, Musa acuminata, Nelumbo nucifera, Nicotiana sp., Phoenix dactylifera, Phaseolus vulgaris, Populus sp., Prunus mume, Pyrus sp., Ricinus communis, Sesamum indicum, Solanum sp., Spinacia oleracea, Tarenaya hassleriana, Theobroma cacao, Vigna angularis, Vitis vinifera, Ziziphus jujuba
Species of your interest not listed? Contact usNot reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
- Application example
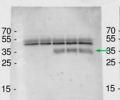
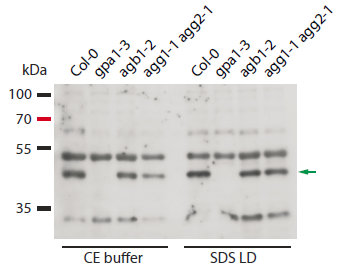 The samples extracted either directly with SDS loading buffer (50mM Tris-HCl pH6.8, 100mM DTT, 2%SDS, 10% glycerol, 0.025% bromophenol blue) and then denatured at 95°C for 5 min, or first extracted with CE buffer (250mM sucrose, 100mM HEPES-KOH pH 7.5, 5% glycerol, 1mM Na2MoO4 x 2H2O, 25mM NaF, 10mM EDTA, 1mM DTT, 0.5%Triton X-100, protease inhibitor cocktail) and then denatured with SDS loading buffer at 70°C for 5 min. 10 µl of proteins were loaded into each well and separated on 10% SDS-PAGE and blotted 1h to PVDF using tank transfer. Blots were blocked with Tris-buffered saline containing 0.05% Tween-20 (TBS-T) and 5% skimmed milk powder for 1h at room temperature (RT) with agitation. The blot was incubated in the primary antibodies indicated at a dilution of 1: 5 000 overnight at 4°C with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed 5 times for 15 min in TBS-T with milk powder at RT with agitation. Blot was incubated in secondary antibody (Goat-Anti-Rabbit AP conjugate, Sigma) diluted 1:5000 for 2h at RT with agitation. The blot was washed as above with TBS-T without milk powder, equilibrated in AP buffer (100mM TRIS pH=9.5, 100mM NaCl, 50mM MgCl2) and then developed with chromogenic detection system, before exposure to a CEA RP NEW film for 20 s.
The samples extracted either directly with SDS loading buffer (50mM Tris-HCl pH6.8, 100mM DTT, 2%SDS, 10% glycerol, 0.025% bromophenol blue) and then denatured at 95°C for 5 min, or first extracted with CE buffer (250mM sucrose, 100mM HEPES-KOH pH 7.5, 5% glycerol, 1mM Na2MoO4 x 2H2O, 25mM NaF, 10mM EDTA, 1mM DTT, 0.5%Triton X-100, protease inhibitor cocktail) and then denatured with SDS loading buffer at 70°C for 5 min. 10 µl of proteins were loaded into each well and separated on 10% SDS-PAGE and blotted 1h to PVDF using tank transfer. Blots were blocked with Tris-buffered saline containing 0.05% Tween-20 (TBS-T) and 5% skimmed milk powder for 1h at room temperature (RT) with agitation. The blot was incubated in the primary antibodies indicated at a dilution of 1: 5 000 overnight at 4°C with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed 5 times for 15 min in TBS-T with milk powder at RT with agitation. Blot was incubated in secondary antibody (Goat-Anti-Rabbit AP conjugate, Sigma) diluted 1:5000 for 2h at RT with agitation. The blot was washed as above with TBS-T without milk powder, equilibrated in AP buffer (100mM TRIS pH=9.5, 100mM NaCl, 50mM MgCl2) and then developed with chromogenic detection system, before exposure to a CEA RP NEW film for 20 s.
Courtesy of Dr. Elena Petusching, Georg-August-University Goettingen, Germany
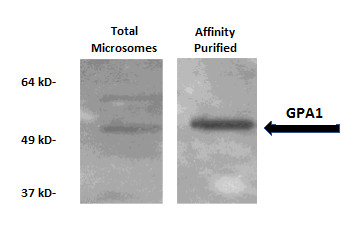
Immunodetection of GPA1 (alpha subunit of heterotrimeric G protein). Microsomes were affinity purified to isolate palmitoylated proteins using the acyl biotin exchange method1. Proteins were separated by SDS-PAGE and immunodetected with anti-GPA1. GPA1 is expected to be modified by S-acylation and was enriched in the affinity-purified fraction.
1. Wan, J., Roth, A. F., Bailey, A. O. & Davis, N. G. Palmitoylated proteins: purification and identification. Nat. Protoc. (2007). doi:10.1038/nprot.2007.225
Palmitoylated (S-acylated) proteins were affinity purified from Arabidopsis thaliana using the acyl biotin exchange method1 and separated using SDS-PAGE. Proteins were electro-transferred to PVDF membrane (Millipore, Cat. No. IVPH00010). The membrane was blocked with 5% non-fat dry milk (NFDM) in Tris-buffered saline (TBS) for two hours at room temperature. All incubations were performed with gentle agitation. The membrane was treated with primary antibody (anti-GPA1 [Agrisera, Cat. No. AS12 2370]; 1:7500 in TBS containing 5% NFDM and 0.05% Tween-20) for 2 hours. Blots were quickly rinsed twice with TBS containing 0.05% Tween (TBS-T), then washed five times for 15 minutes each with TBS-T on a shaker. The membrane was treated for 45 minutes with HRP-tagged secondary antibody (Agrisera, Cat. No. AS09 602) diluted 1:10 000 in TBS-T containing 5% non-fat dried milk. Blots were washed 2-3 times for 15 minutes with TBS-T, followed by two 15-minute washes with TBS. Immunolabeled proteins were detected with chemiluminescent detection reagent and imaged using X-ray film.
Courtesy of John McLarney under the guidance of Dr. Estelle Hrabak, University of New Hampshire, USA - Background
-
Background: GPA1 (G PROTEIN ALPHA-1) is involved as a modulator or transducer in various transmembrane signaling systems. Together with GCR1, may regulate the cell cycle via a signaling cascade Promotes abscisic acid (ABA) responses in guard cells. But, together with GCR1 and GB1, acts as a negative regulator of ABA during seed germination and early seedling development. Involved in the blue light (BL) signaling. Alternative name: GP-alpha-1. - Product Citations
-
Selected references: Liu et al. (2026). The G protein-casein kinase 2 module acts as a pivot in plasma membrane-to-nucleus CLAVATA signaling to control shoot apical meristem size. Nat Commun . 2026 Apr 30. doi: 10.1038/s41467-026-72418-4. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts - Reviews:
-
This product doesn't have any reviews.