Anti-IAA | Indole 3 acetic acid
AS06 193 | Clonality: Polyclonal | Host: Rabbit | Reactivity: IAA | Indole 3 acetic acid
Sold out. Alternative antibodies: AS09 421 and AS09 445
- Product Info
-
Immunogen: BSA-conjugated synthetic indole 3 acetic acid
Host: Rabbit Clonality: Polyclonal Purity: Total IgG Format: Lyophilized in PBS, Phosphate 10 mmol/l; NaCl 150 mmol/l at pH 7.35 Quantity: 1 mg Reconstitution: For reconstitution add 50 µl of sterile water and 50 µl of glycerol Storage: Store lyophilized/reconstituted at -20°C; this aliquote can be freezed and thawed for up to five times and showed stability for at least 2 years, Please, remember to spin tubes briefly prior to opening them to avoid any losses that might occur from lyophilized material adhering to the cap or sides of the tubes Tested applications: ELISA (ELISA), Immunolocalization (IL) Recommended dilution: 1 : 5000-1 : 10 000 (ELISA), 1 : 100-1 :600 (IL) - Reactivity
-
Confirmed reactivity: Arabidoppsis thaliana, Drosera capensis, Euphorbia pulcherrima, Medicago sativa (nodules) Predicted reactivity: Dicots
Species of your interest not listed? Contact usNot reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
Application example
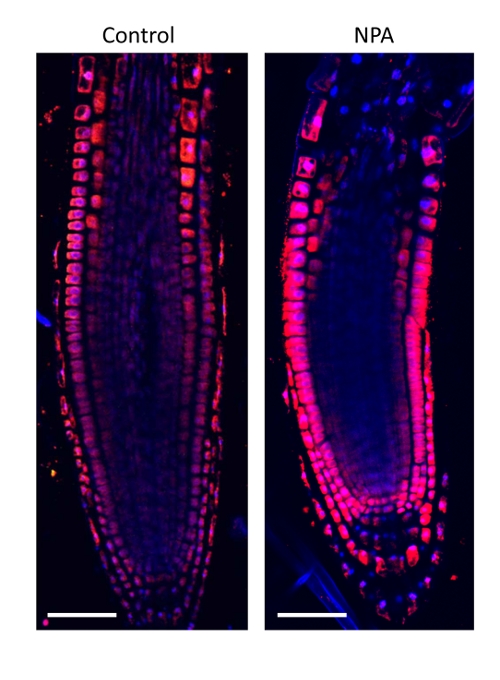
4 days old Arabidopsis thaliana seedlings were treated with 1µM NPA for 24 hours. Seedlings were fixed for 20 min in 4% EDAC in MTSB, and next 30 minutes in 4 % EDAC+ 2% Formaldehyde. Anti-auxin anti-rabbit primary antibody (Agrisera, catalog number AS06 193) diluted 1: 600 plus Goat anti-rabbit IgG (H&L), DyLight® 549 Conjugate (AS11 1815) as secondary antibody diluted in 1: 3000 (shown in red color) were used.
Courtesy Dr. Taras Pasternak, Freiburg University, Germany.
Application examples: 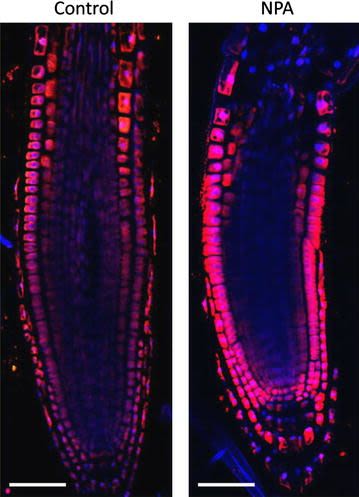
Reactant: Arabidopsis thaliana (Thale cress)
Application: Immunohistochemistry-immunofluorescence
Pudmed ID: 26516341
Journal: Plant Methods
Figure Number: 8A
Published Date: 2015-10-31
First Author: Pasternak, T., Tietz, O., et al.
Impact Factor: 5.139
Open PublicationAuxin immunolocalisation in Arabidopsis roots. Four days old Arabidopsis seedlings were treated with 1 µM 1-N-Naphthylphthalamic acid (NPA) for 24 h to enhance accumulation of auxin in roots. Seedlings were fixed for 20 min in 4 % EDAC in 1× MTSB, and next 30 min in 4 % EDAC+ 2 % Formaldehyde. Anti-indole 3 acetic acid (IAA) rabbit primary antibody (Agrisera, AS06 193) diluted 1:600 plus Goat anti-rabbit IgG (H&L), DyLight® 549 Conjugate (AS09 633) as secondary antibody diluted in 1:3000 (shown in red color) were used. Scale bar 20 µm
- Additional Information
-
Additional information: Total IgG was purified by ammonium sulfate precipiation Additional information (application): For detailed immunolocalization protocol check here.
Quantification of IAA in ELISA using this antibody is described in Bianco and Defez (2009).
Whole-mount immunolocalization is described in Lu et al. (2015). - Background
-
Background: Indole 3 acetic acid (IAA) is the principal auxin in higher plants. This hormone is produced in in cells in the apex and young leaves of a plant. Plant cells synthesize IAA from tryptophan. Different effects caused by auxines include: induction of cell elongation and cell division and have a subsequent results for plant growth and development.
- Product Citations
-
Selected references: La Porta et al. (2019). Metamaterial architecture from a self-shaping carnivorous plant. Proc Natl Acad Sci U S A. 2019 Sep 17;116(38):18777-18782. doi: 10.1073/pnas.1904984116.
Kućko et al. (2019). Spatio-temporal IAA gradient is determined by interactions with ET and governs flower abscission. J Plant Physiol. 2019 Mar 2;236:51-60. doi: 10.1016/j.jplph.2019.02.014.
Nishimura and Koshiba (2019). Immunolocalization of IAA Using an Anti-IAA-C-Antibody Raised Against Carboxyl-Linked IAA. Phototropism. Methods in Molecular Biology, vol 1924. Humana Press, New York, NY.
Lu et al. (2015). OsPIN5b modulates rice plant architecture and yield by changing auxin homeostasis, transport and distribution. Plant J. 2015 Jul 25. doi: 10.1111/tpj.12939.
Bianco and Defez (2009). Medicago truncatula improves salt tolerance when nodulated by an indole-3-acetic acid-overproducing Sinorhizobium meliloti strain.J Exp. Bot. 60, No. 11: 3097-3107. - Protocols
-
Method description for immunolocalization studies
Plant tissue preparation
To examine distribution of IAA, plant materials were embedded in paraplast as described in Lee et al. 2008. The plant materials were cut into small pieces (2 to 3 mm-thick) and immediately fixed in 4% paraformaldehyde in sodium phosphate buffer and 0.1% (v/v) Tween 20 under vacuum for 1 h, and left overnight at 4°C. After fixation, the plant materials were washed in 0.85% (w/v) saline, dehydrated through a graded alcohol series and embedded in paraplast. The plant materials embedded in paraplast sections (10 µm thick) were collected on poly-L-lysine coated slides.Imunolabeling
The plant materials embedded in paraplast sections (10 µm thick) were deparaffinized using histoclear (Cell path, England), rehydrated with an ethanol series, and washed with water. Dewaxed and re-hydrated sections were incubated in 10 mM phosphate-buffered saline (PBS) for 5 min.Blocking
was done using 10 mM PBS, 0.1% (v/v) Tween 20, 1.5% (v/v) Glycine, and 5% (w/v) BSA for 45 min at 20°C.Wash
in 10 mM PBS, 0.88% (w/v) NaCl, 0.1% (v/v) Tween 20, and 0.8% (w/v) BSA) for 5 min and brief wash in10 mM PBS and 0.8% (w/v) BSA.Primary antibody incubation
Agrisera anti-IAA antibody has been used on each slide (100 µl) in dilution 1: 100 and incubated in a humid chamber during overnight at 20ºC.Wash
Two times vigorous wash in 10 mM PBS, 2.9% (w/v) NaCl, 0.1% (v/v) Tween 20, and 0.1% (v/v) BSA for 10 min each were followed by a 10 min wash with 10 mM PBS, 0.88% (w/v) NaCl, 0.1% (v/v) Tween 20, and 0.8% (w/v) BSA and brief rinse with 10 mM PBS and 0.8% (v/v) BSA.Secondary antibody incubation and reaction development
100µl anti-rabbit IgG-alkaline phosphatase-conjugate (Sigma, USA) with 1:100 (w/v) dilution were added to each slide as secondary antibody and incubated overnight in a humidity chamber at 20°C. Two 15 min washes with 10 mM PBS, 0.88% (w/v) NaCl, 0.1% (v/v) Tween 20, and 0.8% (w/v) BSA were followed by a 15 min wash with water. The sections were visualized by applying 200 µl ready-to-use Western Blue (Promega, USA) and incubated in the dark for 15 to 30 min at room temperature (20°C). When blue/purple color was observed, slides were finally washed in water and then dehydrated in ethanol series. Slides were mounted with Depex (BDH, USA). Photographs were taken using Leitz microscope using bright field optics (Leitz, Germany). Negative control was done ommition of primary antibody.
Courtesy Dr LeeExample of Western blot protocol : Membrane blocking, antibodies incubations and detection
1) Saturate the blot membrane with TBS + 5% Blocker for 1 hour at 37°C while mixing
2) Wash the membrane twice for 5 minutes in TBS Tween at 37°C
3) Incubate the membrane with anti-IAA antibodies diluted 1 : 1000 – 1 : 2000 in TBS 0.5% Blocking solution for 2 hours at 37°C
4) Wash the membrane three times for 5 minutes in TBS Tween at 37°C
5) Incubate with a biotinylated secondary antibody diluted (1: 1000-1: 2000) in TBS 0.5% Blocking solution for 2 hours at 37°C
6) Wash the membrane three times for 5 minutes in TBS Tween at 37°C
7) Incubate with Streptavidin-HRP 1μg/ml in TBS 0.5% Blocking solution for 2 hours at room temperature
8) Wash the membrane three times for 5 minutes in TBS at 37°C
9) Incubate in TBS (200ml) + (50mg DAB in 25ml methanol) + (150mg 4-chloro-1-naphtol in 25ml methanol) + 50μl H2O2 30% for a maximum of 30 minutes in the dark
10) Stop the reaction by addition of distilled waterBlocking solution = skim milk (Biorad 170-6404)
TBS = 20mM Tris base, 0.5M NaCl, pH 7.5
TBS Tween = TBS + 0.05% Tween 20
- Reviews:
-
This product doesn't have any reviews.