1
Anti-LEA4-5 | Late embryogenesis abundant protein 4-5 (affinity purified)
AS13 2758A | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana
- Product Info
-
Immunogen: recombinant LEA4-5 from Arabidopsis thaliana: UniProt: Q9FG31, TAIR: AT5G06760, a group 4 LEA protein (Battaglia et al., 2008)
Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 1000 (WB) Expected | apparent MW: 16 kDa
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana Predicted reactivity: Arachis hypogaea, Brassica sp., Glycine max, Medicago truncatula, Phaseolus vulgaris, Thellungiella halophila
Species of your interest not listed? Contact usNot reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
Application information
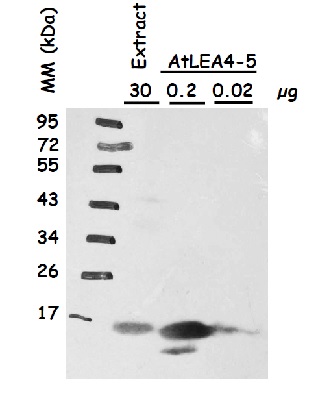
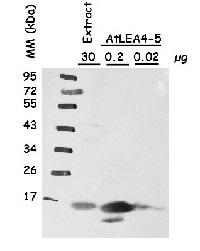
Protein extracts were obtained from Arabidopsis thaliana seeds (extract), beside recombinant AtLEA4-5 loaded in indicated amounts. Following extraction buffer was used: 0.7 M sucrose, 0.5 M Tris-base, 30 mM HCl, 50 mM EDTA, 0.1 M KCl, 2% β-mercaptoethanol, 12 mg/ml poly-vinyl-poly-pyrrolidone (PVPP). This buffer was complemented with 2mL of equilbrated phenol before extraction. Samples were centrifuged and the protein phase was recovered; the extracted proteins were precipitated with 0.1 m ammonium acetate dissolved in methanol, centrifuged and the pellet washed with cold (-20°C) 80% acetone. The protein pellet was dissolved in SDS-solubilization buffer (1% CHES, 2% SDS, 2% ß-mercaptoethanol, 10% glycerol). Thirty µg of seed protein extract was denatured with Laemmli buffer at 95 °C for 5 min and proteins were separated on 15% SDS-PAGE and blotted 1.5 h to nitrocellulose membrane in a liquid transfer system. Blots were blocked with 2% skim milk ON at 4°C with agitation. After rinsing with TBS, blots were incubated in the primary antibody at a dilution of 1:1 000 (anti-AtLEA4-5) for 3h at 4°C with agitation. The antibody solution was decanted and the blot was rinsed briefly, then washed once for 15 min and 3 times for 5 min in TBS-T at 4°C with agitation. Then, blots were incubated in secondary antibody (anti-rabbit IgG horse-radish peroxidase conjugate) diluted to 1:25 000 in for 1h at RT with agitation. The blot was washed as above and developed for 2 min with chemiluminescent detection reagent. Exposure time was for 30 seconds.
Courtesy of Dr. Alejandra Covarrubias, UNAM, Mexico Courtesy of Dr. Alejandra A. Covarrubias, UNAM, Mexico
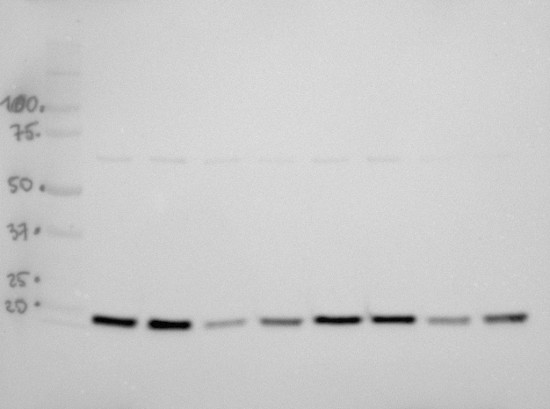
30 μg of total seed protein isolated from Arabidopsis thaliana dry seeds and seeds germinating for 24h, extracted with a buffer containing urea, thiourea, CHAPS and Triton X-100 (Heidorn-Czarna et al., 2018) were denaturated with Laemmli buffer at 95°C for 5 min and separated on 12% SDS-PAGE. Afterwards the gel was blotted for 30 min to PVDF membrane using semi-dry transfer (Trans-Blot Turbo, Bio-Rad). The efficiency of transfer was confirmed by staining of the membrane with Coomassie solution. Blot was blocked with 5% milk in TBS-T for 1h at RT with agitation. Blot was incubated in the primary antibody (anti-LEA4-5, AS13 2758A) at a dilution 1:1000 in 5% milk in TBS-T for 1.5h /RT with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 2 times for min in TBS-T at RT with agitation. Blot was incubated in Agrisera secondary antibody (goat anti-rabbit IgG, HRP-conjugated, AS09 602) diluted to 1:20 000 in 5% milk in TBS-T for 1h/RT with agitation. The blot was washed as above and developed with chemiluminescent detection reagent, using GBox imager (Syngene). Exposure time was: 3 minutes.Analyzed samples:
- Wild-type, dry seeds, I biological replicate
- mutant, dry seeds, I biological replicate
- Wild-type germinating for 24h, I biological replicate
- mutant germinating for 24h, I biological replicate
- Wild-type, dry seeds, II biological replicate
- mutant, dry seeds, II biological replicate
- Wild-type germinating for 24h, II biological replicate
- mutant germinating for 24h, II biological replicate
Courtesy Dr Małgorzata Heidorn-Czarna, University of Wrocław, Poland - Background
-
Background: LEA (Late embryogenesis abundant) proteins are very hydrophilic proteins, described over 25 years ago as accumulating during late stages of plant seed development. Found in vegetative plant tissues following exposure to environmental stress. Synonymes: Putative late embryogenesis abundant protein LEA.
- Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts - Reviews:
-
This product doesn't have any reviews.



