1
Anti-slr1641 | ATP-dependent chaperone clpB
AS08 344 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Cyanobacteria
- Product Info
-
Immunogen: recombinant clpB1 protein, derived from Synechocystis PCC 6803 strain slr1641 sequence; protein has an internal translation site. The nomenclature used is reverse of what is mentioned in the cyanobase.
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 100 µl Reconstitution: For reconstitution add 100 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 3000 (WB) Expected | apparent MW: 98.1 | 85.4 and 105 | 95 kDa for Synechocystis
- Reactivity
-
Confirmed reactivity: Synechocystis PCC 6803, Solanum lycopersicum Predicted reactivity: Cyanobacteria, Francisella sp.
Species of your interest not listed? Contact usNot reactive in: Chlamydomonas reinhardtii - Application Examples
-
Application example
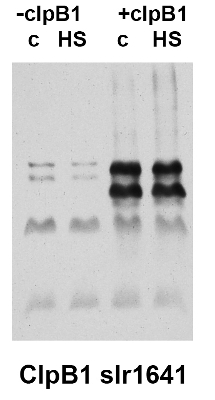
10 μg of total protein from Synechocystis PCC 6803 wild type (+ClpB1) and slr1641 deletion mutant, control (C) and heat shocked samples (HS) was separated on 8% PAA gel and blotted on nitrocellulose membrane . Filters were blocked (1h), incubated with 1: 3000 anti-ClpB1 antibodies (2h) followed by incubation with 1: 2500 secondary anti-rabbit (1h) coupled to HRP and visualization with chemiluminescent detection reagent.
Courtesy of Dr. Elizabeth Vierling, University of Massachusetts, USA - Background
-
Background: ClpB protein is essential for resistance to high temperature stress. It functions to dissolve inactive protein aggregates that accumulate at high temperatures. Giese and Vierling (2002) Changes in oligomerization are essential for the chaperone activity of a small heat shock protein in vivo and in vitro. J Biol Chem; 277(48): 46310-8.
- Product Citations
-
Selected references: Gonzalez-Esquer and Vermaas (2013). ClpB1 overproduction in Synechocystis sp. strain PCC 6803 increases tolerance to rapid heat shock. Appl Environ Microbiol. 2013 Oct;79(20):6220-7. doi: 10.1128/AEM.01661-13. Epub 2013 Aug 2.
- Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Oxygenic photosynthesis poster by prof. Govindjee and Dr. Shevela
Z-scheme of photosynthetic electron transport by prof. Govindjee and Dr. Björn and Dr. Shevela - Reviews:
-
Dr. Rena Gorovits | 2009-04-30Excellent signal in tomato protein extracts



