1
Anti-SHMT | Serine hydroxymethyltransferase
AS05 075 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, Citrus sp., G. max, P. sativum, S. oleracea, V. faba | cellular[compartment marker] of mitochondrial matrix
- Product Info
-
Immunogen: Purified SHMT protein from Spinacia oleracea
Host: Rabbit Clonality: Polyclonal Purity: Total IgG. Protein G purified in PBS pH 7.4. Format: Lyophilized Quantity: 0.5 mg Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1: 1000 to 1 : 5 000 (WB) Expected | apparent MW: 53 kDa (Arabidopsis thaliana)
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Brassica oleracea, Citrus sp., Glycine max, Oryza sativa, Pisum sativum, Spinacia oleracea, Vicia faba
Predicted reactivity: Chlamydomonas reinhardii, Citrus unshiu, Hordeum vulgare, Malus domestica, Medicago truncatula, Nicotiana tatenuata, Populus balsamifera, Ricinus communis, Solanum tuberosum,Theobroma cacao, Vitis vinifera
Species of your interest not listed? Contact usNot reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
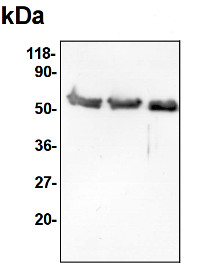
Application example 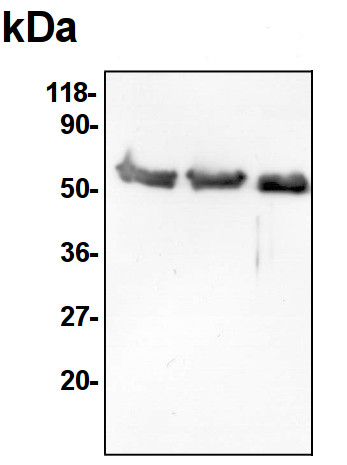
30 µg of total protein from cauliflower Brassica oleracea var. botrytis mitochondrial fraction, extracted according to Rurek el al. 2015 and denatured with standard 1 x conc. Sample Buffer with 2-mercaptoethanol at 80°C for 10 min were separated on 12 % SDS-PAGE and blotted 1h to Immobilon-P using semi-dry transfer in the standard transfer buffer (20 % methanol, 48 mM Tris, 39 mM glycine, 0.0375 % SDS). Blots were blocked with 5 % skimmed milk in PBS-T with 0.01 % Tween20 for 1h at room temperature (RT) with agitation. Blot was incubated in the primary antibody in 2 % skimmed milk in PBS-T in 2 % skimmed milk at a dilution of 1: 10 000 for 1h at RT with agitation in TBS-T. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in TBS-T at RT with agitation. Blot was incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated, AS09 602, Agrisera ) diluted to 1:10 000 in PBS-T with 2 % milk, for 1h at RT with agitation. The blot was washed as above and developed for min with enhanced chemiluminescence. Exposure time was 5 s.
Courtesy of Michal Rurek, D.Sc., Adam Mickiewicz University, Poznań (Poland)Application examples: 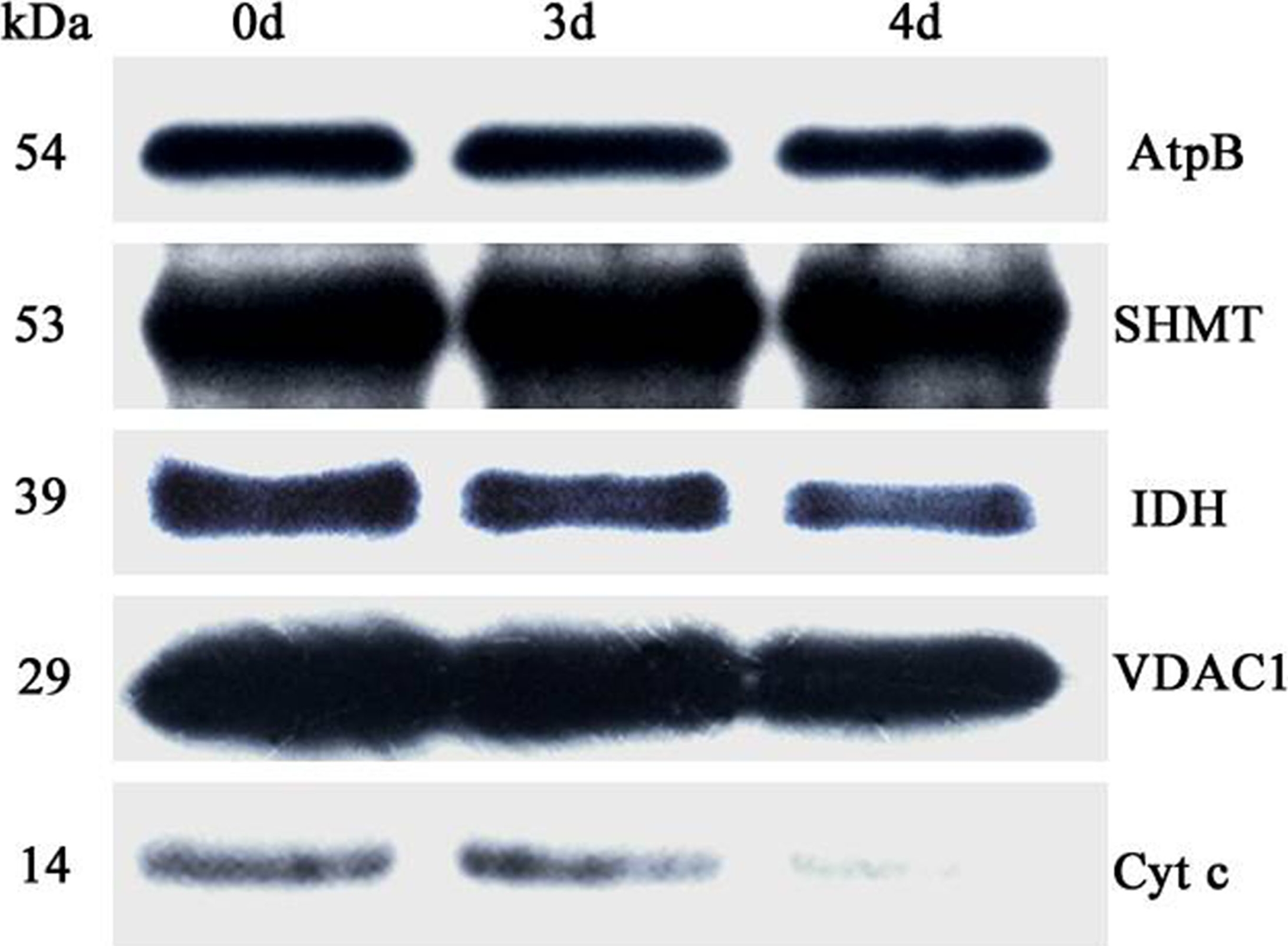
Reactant: Oryza sativa (Asian rice)
Application: Western Blotting
Pudmed ID: 27124767
Journal: PLoS One
Figure Number: 6A
Published Date: 2016-04-29
First Author: Yin, G., Whelan, J., et al.
Impact Factor: 2.942
Open PublicationAbundance of a variety of known mitochondrial proteins by the immunoblot analysis of mitochondria isolated from 0 d, 3 d, and 4 d aged rice embryos after 48 h imbibition.Total 10 ?g protein was separated by SDS gel electrophoresis and blotted to supported polyvinylidene difluoride, then probed with antibodies against beta subunit of ATP synthase (AtpB), serine hydroxymethyltransferase (SHMT), isocitrate dehydrogenase (IDH), voltage dependent anion channel 1 (VDAC1) and cytochrome c (Cyt c).
- Additional Information
-
Additional information (application): This antibody can be used on total leaf extracts and isolated mitochondria
- Background
-
Background: Serine hydroxymethyltransferase (SHMT) is part of the mitochondrial enzyme complex.
- Product Citations
-
Selected references: Hoa et al. (2024). Proteomic analysis on symbiotic differentiation of mitochondria in soybean nodules. Comparative Study Plant Cell Physiol. 2004 Mar;45(3):300-8. doi: 10.1093/pcp/pch035.
Oikawa et al. (2022) Pexophagy suppresses ROS-induced damage in leaf cells under high-intensity light. Nat Commun. 2022;13(1):7493. Published 2022 Dec 5. doi:10.1038/s41467-022-35138-z
Li et al. (2021) Isolation and comparative proteomic analysis of mitochondria from the pulp of ripening citrus fruit. Hortic Res. 2021 Feb 1;8(1):31. doi: 10.1038/s41438-021-00470-w. PMID: 33518707; PMCID: PMC7848011.
Guo et al. (2021) The pentatricopeptide repeat protein GEND1 is required for root development and high temperature tolerance in Arabidopsis thaliana,Biochemical and Biophysical Research Communications,Volume 578,2021,Pages 63-69,ISSN 0006-291X,https://doi.org/10.1016/j.bbrc.2021.09.022.(https://www.sciencedirect.com/science/article/pii/S0006291X21013164)
Rurek et al. (2018). Mitochondrial Biogenesis in Diverse Cauliflower Cultivars under Mild and Severe Drought. Impaired Coordination of Selected Transcript and Proteomic Responses, and Regulation of Various Multifunctional Proteins, Int. J. Mol. Sci. 2018, 19(4), 1130; doi:10.3390/ijms19041130 (Special issue: Plant Mitochondria)
Rurek et al. (2018). Cold and Heat Stress Diversely Alter Both Cauliflower Respiration and Distinct Mitochondrial Proteins Including OXPHOS Components and Matrix Enzymes, Int. J. Mol. Sci. 2018, 19(3), 877; doi:10.3390/ijms19030877 (Special issue: Plant Mitochondria)
Yin et al. (2016). Comprehensive Mitochondrial Metabolic Shift during the Critical Node of Seed Ageing in Rice. PLoS One. 2016 Apr 28;11(4):e0148013. doi: 10.1371/journal.pone.0148013. eCollection 2016.
Long et al. (2015). Contributions of photosynthetic and non-photosynthetic cell types to leaf respiration in Vicia faba L. and their responses to growth temperature. Plant Cell Environ. 2015 Apr 1. doi: 10.1111/pce.12544.
Wei et al. (2013). Folate polyglutamylation eliminates dependence of activity on enzyme concentration in mitochondrial serine hydroxymethyltransferases from Arabidopsis thaliana. Arch Biochem Biophys 1;536(1):87-96.
Camejo et al. (2012).Salinity-inducedchanges inS-nitrosylation ofpeamitochondrialproteins. J. Proteomics Dec.11. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts - Reviews:
-
Micha? RUREK | 2014-02-145 µg of mitochondrial proteins from cauliflower (Brassica oleracea var. botrytis) curds (apical layer) was separated on 12 % SDS-PAGE (Laemmli- type) and blotted 1h to Immobilone P (Millipore) using Sedryt apparatus (Kucharczyk). After blocking (5% milk in PBS-T) for 1 h at RT, blots were incubated in the primary antibody at a dilution of 1: 10000 for 1h at room temperature with agitation.The primary antibody, diluted in 2% milk in PBS-T was reused several times. Secondary, anti-rabbit HRP- linked antibodies were bound at 1/100000 dilution. Single band of 55 kDa was detected using standart GE Healthcare ECL reagents.
Accessories

AS05 074 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana,P.hybrida cv. Mitchell, P. grandiflora, S. oleracea, T. aestivum, V. faba | cellular [compartment marker] of mitochondrial matrix
AS06 203A | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, C.annuum, G. max, L. esculentum, O. sativa, P. sativum, S. tuberosum, Z. mays | cellular [compartment marker] of mitochondrial matrix
AS07 212 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, A. palmeri, S. oleracea, di and monocots | Cellular [compartment marker] of mitochondrial outer membrane

