1
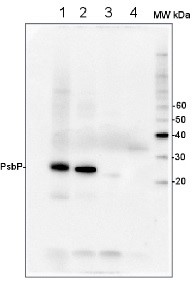
Anti-PsbP | 23 kDa protein of the oxygen evolving complex (OEC) of PSII (anti-protein)
AS06 142-23 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, Ch. reinhardtii, P. banksiana. , S.oleracea, T. aestivum
- Product Info
-
Immunogen: native, purified 23 kDa protein from Spinacia oleracea,UniProt: P12302
Host: Rabbit Clonality: Polyclonal Purity: Total IgG. Protein G purified in PBS pH 7.4. Format: Lyophilized Quantity: 200 µg Reconstitution: For reconstitution add 200 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 2000-1 : 5000 (WB) Expected | apparent MW: 28 | 23 kDa
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Chlamydomonas reinhardti, Hordeum vulgare, Pinus banksiana, Spinacia oleracea, Triticum aestivum Predicted reactivity: Musa acuminata, Oryza sativa, Pinus monticola, Pisum sativum, Populus balsamifera, Solanum lycopersicum
Species of your interest not listed? Contact usNot reactive in: Chlorella vulgaris, Synechococcus sp. PCC 7942
- Application Examples
-
Application example
2 µg of total protein from Arabidopsis thaliana leaf (1) Hordeum vulgare leaf (2), Chlamydomonas reinhardtii total cell (3), Synechococcus sp. 7942 total cell (4), all extracted with PEB (AS08 300) and separated on 4-12% NuPage (Invitrogen) LDS-PAGE and blotted 1h to PVDF. Blots were blocked immediately following transfer in 2% blocking reagent in 20 mM Tris, 137 mM sodium chloride pH 7.6 with 0.1% (v/v) Tween-20 (TBS-T) for 1h at room temperature with agitation. Blots were incubated in the primary antibody at a dilution of 1: 10 000 for 1h at room temperature with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in TBS-T at room temperature with agitation. Blots were incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated) diluted to 1:50 000 in 2% blocking solution for 1h at room temperature with agitation. The blots were washed as above and developed for 5 min with chemiluminescent detection reagent according the manufacturers instructions. Images of the blots were obtained using a CCD imager (FluorSMax, Bio-Rad) and Quantity One software (Bio-Rad).
- Additional Information
-
Additional information (application): Load per well on cell extract of Pinus banksiana (Jack Pine) was 7 µg.
This antibody can be used as a loading control for Chlamydomonas reinhardtii while it not so suitable for higher plants as accumulation of these proteins might drop to 12.5-25 % of the WT level in mutants defective for PSII core (Schult et al. 2007). - Background
-
Background: PSII reaction centre components are generating the redox potential required to drive highly oxidizing water splitting reaction. Four Mn atoms are present on a lumenal surface and form the catalyctic site of the water-splitting reaction which is in close association with the 33 kDa (PsbO), 23 kDa (PsbP) and 17 kDa (PsbQ) extrinistic subunits of oxygen evolving complex OEC. A 33-kDa extrinsic protein is also termed the Mn-stabilizing protein (MSP), however recent evidences shown that it is C-terminal domain of PsbA (D1) protein which is involved in in the assembly and stabilization of the OEC.
- Product Citations
-
Selected references: Collombat et al. (2025). Arabidopsis conditional photosynthesis mutants abc1k1 and var2 accumulate partially processed thylakoid preproteins and are defective in chloroplast biogenesis. Commun Biol . 2025 Jan 22;8(1):111. doi: 10.1038/s42003-025-07497-y.
Mu et al. (2024). Plastid HSP90C C-terminal extension region plays a regulatory role in chaperone activity and client binding.Plant J. 2024 Jul 5.doi: 10.1111/tpj.16917.
Vidal-Meireles, et al. (2023). The lifetime of the oxygen-evolving complex subunit PSBO depends on light intensity and carbon availability in Chlamydomonas. Plant Cell Environ. 2023;46(2):422-439. doi:10.1111/pce.14484
Lim et al (2022) Arabidopsis guard cell chloroplasts import cytosolic ATP for starch turnover and stomatal opening. Nat Commun. 2022 Feb 3;13(1):652. doi: 10.1038/s41467-022-28263-2. PMID: 35115512; PMCID: PMC8814037.
Cecchin et al (2021) LPA2 protein is involved in photosystem II assembly in Chlamydomonas reinhardtii. Plant J. 2021 Jul 4. doi: 10.1111/tpj.15405. Epub ahead of print. PMID: 34218480.
Jiang et al. (2020). Plastid chaperone HSP90C guides precursor proteins to the SEC translocase for thylakoid transport. J Exp Bot. 2020 Aug 27;eraa399.doi: 10.1093/jxb/eraa399.
Nama et al. (2018). Non-photochemical quenching-dependent acclimation and thylakoid organization of Chlamydomonas reinhardtii to high light stress. Photosynth Res. 2018 Jul 7. doi: 10.1007/s11120-018-0551-7.
Yang-Er Chen et al. (2017). Responses of photosystem II and antioxidative systems to high light and high temperature co-stress in wheat. J. of Exp. Botany, Volume 135, March 2017, Pages 45–55.
Wang et al. (2008). Beta-lactone probes identify a papain-like peptide ligase in Arabidopsis thaliana. Nat Chem Biol. 4: 557-563.
Penzler et al. (2022) Commonalities and specialties in photosynthetic functions of PROTON GRADIENT REGULATION5 variants in Arabidopsis. Plant Physiol. 2022;190(3):1866-1882. doi:10.1093/plphys/kiac365
Cecchin et al (2021) LPA2 protein is involved in photosystem II assembly in Chlamydomonas reinhardtii. Plant J. 2021 Jul 4. doi: 10.1111/tpj.15405. Epub ahead of print. PMID: 34218480.
Loudya et al. (2021) Cellular and transcriptomic analyses reveal two-staged chloroplast biogenesis underpinning photosynthesis build-up in the wheat leaf. Genome Biol. 2021 May 11;22(1):151. doi: 10.1186/s13059-021-02366-3. PMID: 33975629; PMCID: PMC8111775.
Terentyev (2020: The Main Structural and Functional Characteristics of Photosystem-II-Enriched Membranes Isolated From Wild Type and cia3 Mutant Chlamydomonas reinhardtii. Life (Basel). 2020 May 14;10(5):E63. doi: 10.3390/life10050063..
Tang el al. (2020). OsNSUN2-Mediated 5-Methylcytosine mRNA Modification Enhances Rice Adaptation to High Temperature. Dev Cell. 2020 May 4;53(3):272-286.e7. doi: 10.1016/j.devcel.2020.03.009.
Smythers et al. (2019). Characterizing the effect of Poast on Chlorella vulgaris, a non-target organism. Chemosphere Volume 219, March 2019, Pages 704-712.
Vojta and Fulgosi (2019). Topology of TROL protein in thylakoid membranes of Arabidopsis thaliana. Physiol Plant. 2019 Jan 20. doi: 10.1111/ppl.12927.
Da-Wei Yang et al. (2018). Genetically engineered hydrogenases promote biophotocatalysis-mediated H 2 production in the green alga Chlorella sp. DT. Int J of Hydrogen Energy.
Liu et al. (2018). Effects of PSII Manganese-Stabilizing Protein Succinylation on Photosynthesis in the Model Cyanobacterium Synechococcus sp. PCC 7002. Plant Cell Physiol. 2018 Jul 1;59(7):1466-1482. doi: 10.1093/pcp/pcy080.
Glowacka et al. (2018). Photosystem II Subunit S overexpression increases the efficiency of water use in a field-grown crop. Nat Commun. 2018 Mar 6;9(1):868. doi: 10.1038/s41467-018-03231-x.
Du et al. (2018). Galactoglycerolipid Lipase PGD1 Is Involved in Thylakoid Membrane Remodeling in Response to Adverse Environmental Conditions in Chlamydomonas. Plant Cell. 2018 Feb;30(2):447-465. doi: 10.1105/tpc.17.00446.
Sultan et al. (2017). The Reverse Transcriptase/RNA Maturase Protein MatR Is Required for the Splicing of Various Group II Introns in Brassicaceae Mitochondria. Plant Cell. 2016 Nov;28(11):2805-2829.
Gandini et al. (2017). The transporter SynPAM71 is located in the plasma membrane and thylakoids, and mediates manganese tolerance in Synechocystis PCC6803. New Phytol. 2017 Mar 20. doi: 10.1111/nph.14526.
Yang-Er Chen et al. (2017). Responses of photosystem II and antioxidative systems to high light and high temperature co-stress in wheat. J. of Exp. Botany, Volume 135, March 2017, Pages 45–55. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts - Reviews:
-
Soujanya Kuntam | 2021-11-09We used this antibody to detect PsbP in complete protein extracts of C. reinhardtii using western blotting. The antibody was used at 1:1000 dilution and worked well without much optimization needed
Related products
AS06 142-16 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, H. vulgare, P. abies, T. aestivum, Z. mays