1
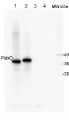
Anti-PsbQ | 16 kDa protein of the oxygen evolving complex (OEC) of PSII
AS06 142-16 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, H. vulgare, P. abies, T. aestivum, Z. mays
- Product Info
-
Immunogen: KLH-conjugated synthetic peptide derived from available PsbQ protein sequences including Arabidopsis thaliana At4g21280.
Peptide used to elicit this antibody is conserved in both isoforms, Arabidopsis thaliana PsbQ1 and PsbQ2.Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 200 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 1000 (WB) Expected | apparent MW: 23.8 | 16 kDa - Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Hordeum vulgare, Picea abies, Pisum sativum, Triticum aestivum, Zea mays Predicted reactivity: Catalpa bungei, Lolium multiflorum, Oryza sativa, Picea sitcHensis, Populus balsamifera, Spinacia oleracea, Triticum aestivum
Species of your interest not listed? Contact usNot reactive in: Chlamydomonas reinhardtii, Physcomitrella patens, Zostera marina
- Application Examples
-
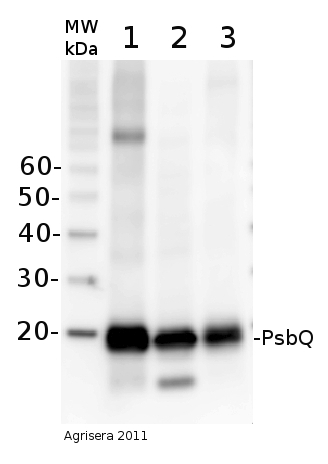
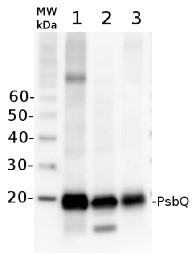
5 µg of total protein from Arabidopsis thaliana (1), Hordeum vulgare (2), Zea mays (3) extracted with Agrisera PEB buffer (AS08 300) were separated on 4-12 % NuPAGE Bis-Tris gel (Invitrogen) and blotted 1h to PVDF. Blots were blocked with blocking reagent for 1.5 h at room temperature (RT) with agitation. Blot was incubated in the primary antibody at a dilution of 1: 10 000 for 1h at RT with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in TBS-T at RT with agitation. Blot was incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated, Agrisera, AS09 602) diluted to 1:25 000 in for 1h at RT with agitation. The blot was washed as above and developed for 5 min with chemiluminescent detection reagent, according to the manufacturers instructions. Exposure time was 44 seconds.Application examples: 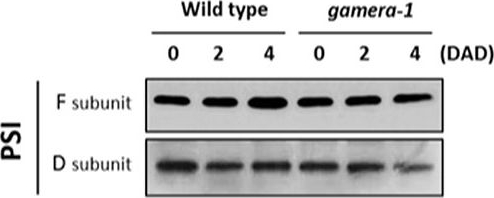
Reactant: Arabidopsis thaliana (Thale cress)
Application: Western Blotting
Pudmed ID: 28791032
Journal: Front Plant Sci
Figure Number: 2A
Published Date: 2017-08-10
First Author: Kohzuma, K., Froehlich, J. E., et al.
Impact Factor: 5.435
Open PublicationChanges in the protein levels of photosynthetic components under extended dark exposure in wild-type and gamera-1. Immunoblot detection of photosynthetic proteins from leaves of Ws and gamera-1 plants incubated after dark adaptation for 0, 2, and 4 days was examined. Specifically, essentially thylakoid fractions were assayed to determine the content of the following proteins: ?-subunit of ATP synthase; the D1 protein, OEC17, OEC23, and OEC33 of PSII; Cyt f and Rieske protein of the cytochrome b6f complex; and the F and D subunits of PSI, after extended dark treatment. Proteins were resolved via SDS-PAGE gel based on equal microgram chlorophyll per lane loading and processed as described in Section “Materials and Methods”. The Large subunit of RuBisco and LHCII stained with either CBB or Ponceau red, respectively, are presented here as loading controls. DAD indicates days after dark adaptation.
- Additional Information
-
Additional information (application): This product can be sold with ProClin if requested
- Background
-
Background: PSII reaction centre components are generating the redox potential required to drive highly oxidizing water splitting reaction. Four Mn atoms are present on a lumenal surface and form the catalyctic site of the water-splitting reaction which is in close association with the 33 kDa (PsbO), 23 kDa (PsbP) and 17 kDa (PsbQ) extrinistic subunits of oxygen evolving complex OEC. A 33-kDa extrinsic protein is also termed the Mn-stabilizing protein (MSP), however recent evidences shown that it is C-terminal domain of PsbA (D1) protein which is involved in in the assembly and stabilization of the OEC. Synonymes: PSBQ, PSBQA
- Product Citations
-
Selected references: Silvestre et al. (2025). A Holistic Investigation of Arabidopsis Proteomes Altered in Chloroplast Biogenesis and Retrograde Signalling Identifies PsbO as a Key Regulator of Chloroplast Quality Control. Plant Cell Environ. 2025 Aug;48(8):6373-6396. doi: 10.1111/pce.15611.
Collombat et al. (2025). Arabidopsis conditional photosynthesis mutants abc1k1 and var2 accumulate partially processed thylakoid preproteins and are defective in chloroplast biogenesis. Commun Biol . 2025 Jan 22;8(1):111. doi: 10.1038/s42003-025-07497-y.
Charras et al. (2024). An efficient protocol for extracting thylakoid membranes and total leaf proteins from Posidonia oceanica and other polyphenol-rich plants. Plant Methods. 2024 Mar 11;20(1):38. doi: 10.1186/s13007-024-01166-7.
Trotti et al. (2024). Physiological Responses to Salt Stress at the Seedling Stage in Wild (Oryza rufipogon Griff.) and Cultivated (Oryza sativa L.) Rice Plants (Basel). 2024 Jan 26;13(3):369. .
Yang-Er Chen et al. (2017). Responses of photosystem II and antioxidative systems to high light and high temperature co-stress in wheat. J. of Exp. Botany, Volume 135, March 2017, Pages 45–55.
Pavlovič et al. (2016). Light-induced gradual activation of photosystem II in dark-grown Norway spruce seedlings. Biochim Biophys Acta. 2016 Feb 18. pii: S0005-2728(16)30028-7. doi: 10.1016/j.bbabio.2016.02.009.
Albanese et al. (2016). Isolation of novel PSII-LHCII megacomplexes from pea plants characterized by a combination of proteomics and electron microscopy. Photosynth Res. 2016 Jan 9.
Grassl et al. (2012). Early events in plastid protein degradation in stay-green Arabidopsis reveal differential regulation beyond the retention of LHCII and chlorophyll. J. Proteome Res. October 2. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts - Reviews:
-
This product doesn't have any reviews.
Related products
AS06 167 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, H. vulgare, N. tabacum, P.abies, P. sativum, P. patens
AS05 059 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, N. tabacum, P. sativum, S. oleracea, T. aestivum
AS06 142-23 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, Ch. reinhardtii, P. banksiana. , S.oleracea, T. aestivum