1
Anti-Cas9 | CRISPR-associated endonuclease 9 (polyclonal)
AS16 3690 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Cas9 from Streptococcus thermophilus
- Data sheet
-
- Product Info
-
Immunogen: KLH-conjugated synthetic peptide derived from Streptococcus thermophilus Cas9/Csn1 protein sequence, UniProt: Q03JI6
(peptide sequence is conserved in pYLCRISPR/Cas9Pubi-H vector)Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Immunolocalization (IL), Western blot (WB) Recommended dilution: 1 : 100 (IL), 1 : 1000 (WB) Expected | apparent MW: Depends on the MW of the protein that is the fusion partner. - Reactivity
-
Confirmed reactivity: Staphylococcus aureus Predicted reactivity: Staphylococcus pyogenes UniProt: Q99ZW2 - Application Examples
-
Application example 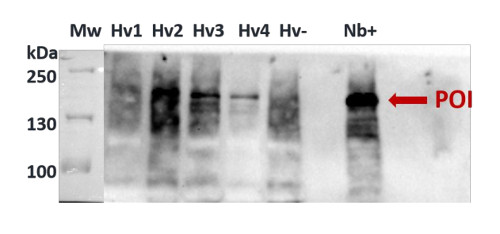
Nicotiana benthamiana (N b.): sample from 5 weeks old plants second leaves (previously infiltrated with A.tumefaciens containing Cas9 transformation casettes(Nb+). Hordeum vulgare (Hv 1-4 , Hv- samples): 2 months old plants’ youngest leaves (stably transformed (Hv 1, -2, -3, -4) and wild type (Hv-) plants). 100 mg plant tissue (homogenised in liquid N2) +400 μl extraction buffer (15 min 4 °C 1500rpm centrifugation). Tissue was extracted with following extaction buffer: 10 mM Tris-HCl, 1 mM EDTA, 150 mM NaCl, 10 V% glicerol, 0,5 V% Nonidet P-40, 5 mM NaF. Right before use: 1 mM DTT, 100 mM PMSF, 200 mM Na3VO4 is added to the extraction buffer. Denaturation: sample in extraction buffer + equal volume 2X Laemmli buffer, 72°C, 10 min.
20 μl of the samples were separated on 8% SDS-PAGE, and blotted with transblot turblot turbo (BioRad) to 0.45 um pore size PVDF (Hybond, GE). Blots were blocked with 5% milk powder in PBST for 60 min at room temperature (RT) with agitation. Blot was incubated in the primary antibody at a dilution of 1: 1000 for 160 min at RT with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed 2 times for 10 min in PBST buffer at RT with agitation. Blot was incubated in secondary antibody (Goat anti-rabbit from Agrisera, AS09 602) diluted to 1:20 000 in PBST for 90 min at RT with agitation. The blot was washed as above and developed for 1 min with Bio-Rad Clarity Western ECL substrate using Bio-Rad ChemiDocTM Imaging System. Exposure time was 170.6 sec.
Courtesy Dr. Éva Hamar, ABC, Hungary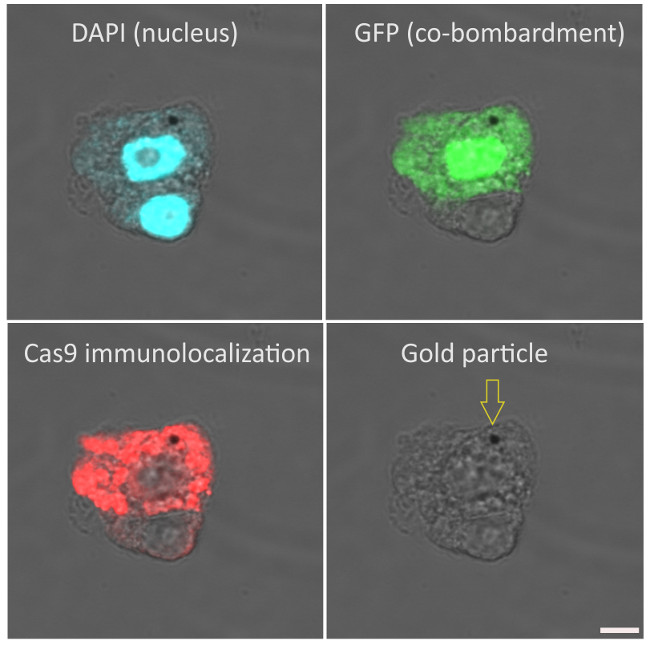
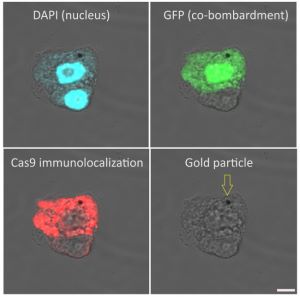
Immunolocalization of Cas9 on maize (Zea mays, cv. H1233) plant suspension cultures. Rabbit anti-Cas9 antibody (Agrisera AS16 3690), diluted 1:100 and DyLight 550 anti-rabbit secondary antibody (Agrisera AS11 1782) diluted 1:300, (red) are used for immunodetection following formaldehyde fixation, cell wall digestion and detergent permeabilization of cells. DAPI is used as nuclear marker (blue). Gold particle used for microprojectile bombardment of plasmids (GFP and Cas9) is indicated with an arrow at the last column. Scalebar is 5µm.
Courtesy of Dr. Ferhan Ayaydin, Hungarian Academy of Science, Hungary
Labeling and detection protocol
Fixation (30 min) 4% paraformaldehyde in PBS (pH 7.4) with 0.01% TritonX-100. (3x5min PBS wash)
Cell wall digestion (40 min): 1% Cellulase, 1% Pectinase in 0.5% (w/v) MES buffer (pH 5.6) (2x5 min PBS wash)
Immobilization of cells (10mins): Cells in PBS were settled onto poly-L-Lysine coated coverslips, excess PBS removed without air drying the cells.
Membrane permeabilization (10 min): 0.5% TritonX-100 in PBS (3x5min PBS wash) Blocking (10mins): 5% Fish gelatin in PBS
Primary antibody incubation (2.5 h at 24°C): Agrisera (AS16 3690) rabbit anti-Cas9 polyclonal antibody diluted 1:100 in blocking buffer (4x5 min blocking buffer wash)
Secondary antibody incubation (1h at 24°C): goat anti-rabbit DyLight 550 antibody (Agrisera AS11 1782) diluted 1:300 in blocking buffer. (3x5min PBS wash)
Nuclear counterstaining (5 min): 200ng/ml DAPI in PBS (brief PBS wash)
Mounting: Fluoromount G mounting medium was used to mount coverslips onto glass slides. Imaging: Olympus FV1000 confocal microscope with 40x (NA1.3) oil immersion objective. - Background
-
Background: Cas9 (CRISPR-associated endonuclease 9) is an RNA-guided DNA endonuclease associated with the Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) type II adaptive immunity system. CRISPR clusters are transcribed and processed into CRISPR RNA (crRNA). Cas9 protein serves as a genome engineering tool to induce site-directed double strand breaks in DNA. - Protocols
-
Antibody protocols
- Reviews:
-
This product doesn't have any reviews.



