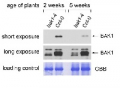
1
Anti-FLS2 | Flagellin-sensitive 2
AS12 1857 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana
- Product Info
-
Immunogen: KLH-conjugated peptide derived from Arabidopsis thaliana FLS2 sequence, UniProt: Q9FL28,TAIR: AT5G46330 Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles,Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 5000 (WB) Expected | apparent MW: 126 kDa (without propeptide) | 175-180 kDa
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana Predicted reactivity: Arabidopsis halleri, Capsella rubella, Glycine max, Hordeum vulgare, Populus trichocarpa, Solanum lycopersivum, Vitis vinifera
Species of your interest not listed? Contact usNot reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
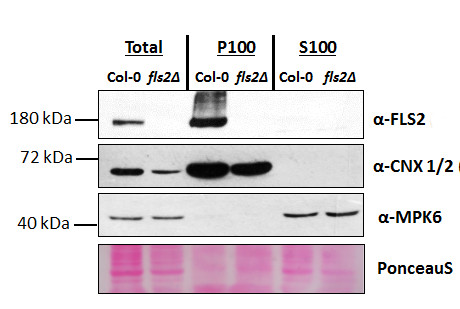
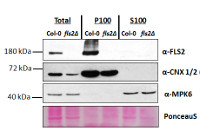
Proteins were isolated from 7-day old Arabidopsis thaliana seedlings, Col-0 (wild-type) and fls2 null mutant (SALK_093905; Heese et al., 2007). Total proteins (T) were isolated and then fractionated by ultracentrifugation at 100,000 x gravity for 30 min at 4°C into soluble (S100) and microsomal (P100) proteins as described in LaMontagne et al. (2016). For each fraction, 30 µg of proteins were denatured at 65°C for 5 min, separated on an 8 % SDS-PAGE and transferred for 1h using a tank transfer system to nitrocellulose membrane. Blots were blocked with 1x PBS (Fisher Scientific BP665-1) + 0.1 %Tween 20 (PBS-T) + 5% milk for 1.5h at room temperature (RT) with agitation. Blots were incubated with primary antibody at a dilution of 1: 1000 overnight at 4°C with agitation in 1x PBS-T + 5% milk. The antibody solution was decanted, and the blot washed four times (1x5min and 3x6min) in 1x PBS-T at RT with agitation. Blot was incubated with secondary antibody [Goat anti Rabbit IgG (H&L) –HRP conjugated; AS09 602) diluted to 1:10 000 in 1x PBS-T + 5% milk for 3h 15min at RT with agitation. The blot was washed as above and developed for 5 min with chemiluminescent detection reagent. Exposure time was 5min. α-CNX 1/2 (AS12 2365, Agrisera) and α-MPK6 (Heese et al., 2007) confirmed separation of microsomal from soluble fraction.
Courtesy of Gayani Ekanayke1, Grant Mc Gowan1,2 & Dr. Antje Heese1 [1 Div. of Biochemistry, Interdisciplinary Plant Group (IPG) - University of Missouri; Columbia, MO, 65211, USA; 2 Dep. of Molecular & Cell Biology, University of Cape Town, Rondebosch 7701, South Africa].
References Heese, A. et al. (2007). The receptor-like kinase SERK3/BAK1 is a central regulator of innate immunity in plants. PNAS. Vol. 104 no. 29 p. 12217–12222.
LaMontagne, E. et al. (2016). Isolation of Microsomal Membrane Proteins from Arabidopsis thaliana. Current Protocols in Plant Biology 1:1-18. doi: 10.1002/cppb.20020.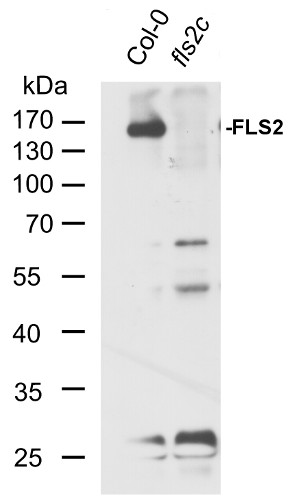
Plant material: Arabidopsis thaliana Col-0 (wild type) fls2c mutant (described in Nekrasov et al. 2009). Method: Two-week-old seedlings (grown under short day conditions, 8h light) were collected and frozen in liquid nitrogen. Approximately 100mg of plant material were extracted in 0.2 ml of homogenization buffer (250 mM sucrose, 50 mM HEPES-KOH pH 7.5, 5% glycerol, 0.5% Triton X-100, 50 mM Na4P2O7, 1 mM Na2MoO4, 25 mM NaF, 2 mM DTT, Sigma plant protease inhibitor cocktail) with a glass pistil and a small amount of sand in an 1.5ml Eppendorf tube. Another 0.8 ml of buffer were added and the extract was mixed thoroughly. Debris was pelleted by centrifugation in a table-top microcentrifuge at 13 000 rpm. Thehe supernatant was mixed with 4x SDS loading buffer (200 mM TRIS-HCl pH 6.8, 400 mM DTT, 8% SDS, 40% glycerol, 0.1% bromophenol blue) and boiled at 95°C for 5min. 10µl were run on an 10% polyacrylamide gel and blotted onto a 0.45µm PVDF membrane (Carl Roth). The membrane was blocked for 1h in TBS-T (150mM NaCl, 10mM Tris-HCl pH8, 0.05% Tween-20) containing 5% skimmed milk powder. The primary antibody (Agrisera Rabbit anti-FLS2, 1µg/µl) was diluted 1:5000 in TBS-T containing 5% milk powder and incubated on the membrane overnight at 4°C. Then the membrane was washed 5 times 15min with TBS-T containing 5% milk powder. The secondary antibody was Agrisera goat-anti rabbit IgG HRP-conjugated (AS09 602) at a dilution of 1:5000 diluted in TBS-T containing 5% milk powder in the same solution and incubated on the membrane at room temperature for 2h. The membrane was then washed 5 times 15min with TBS-T (no milk powder) and the blot was developed using chemiluminescent detection reagent.
Courtesy of Dr. Elena Petutsching, Georg-August-University Goettingen, Germany - Background
-
Background: FLS2 (Flagellin-sensitive 2) is a pattern-recognition receptor and a single-pass membrane protein. Alternative names: FLAGELLIN-SENSITIVE 2, FLAGELLIN-SENSING 2, FLS2, LRR receptor-like serine/threonine-protein kinase FLS2 - Product Citations
-
Selected references: Jo et al. (2026). Shigella Effector VirA Suppresses Plant Immunity by Compromising PRA1.F3-Dependent Accumulation of FLS2 at the Plasma Membrane. Plant Cell Environ . 2026 Apr 14. doi: 10.1111/pce.70541.
Zhang et al. (2026). The RING-Type Ubiquitin Ligase XBAT35. 2 Positively Modulates Plant Immunity by Promoting FLS2 Protein Accumulation. Mol Plant Microbe Interact . 2026 Mar 3. doi: 10.1094/MPMI-02-26-0015-R.
Kalischuk et al. (2022) Amplification of cell signaling and disease resistance by an immunity receptor Ve1Ve2 heterocomplex in plants. Commun Biol. 2022 May 25;5(1):497. doi: 10.1038/s42003-022-03439-0. PMID: 35614138; PMCID: PMC9132969.
Wang et al. (2021) Arabidopsis PUB2 and PUB4 connect signaling components of pattern-triggered immunity. New Phytol. 2021 Dec 17. doi: 10.1111/nph.17922. Epub ahead of print. PMID: 34918346.
Ngou et al. (2021) Mutual potentiation of plant immunity by cell-surface and intracellular receptors. Nature. 2021 Mar 10. doi: 10.1038/s41586-021-03315-7. Epub ahead of print. PMID: 33692545.
Hilleary et al. (2020). Tonoplast-localized Ca2+ pumps regulate Ca2+ signals during pattern-triggered immunity in Arabidopsis thaliana. Proc Natl Acad Sci U S A . 2020 Aug 4;117(31):18849-18857.doi: 10.1073/pnas.2004183117.
Zhang et al. (2019). An important role of L -fucose biosynthesis and protein fucosylation genes in Arabidopsis immunity. New Phytol. 2018 Dec 15. doi: 10.1111/nph.15639.
Yang et al. (2019). A Plant Immune Receptor Degraded by Selective Autophagy. Mol Plant. 2019 Jan 7;12(1):113-123. doi: 10.1016/j.molp.2018.11.011.
Zhang et al. (2018). An important role of L -fucose biosynthesis and protein fucosylation genes in Arabidopsis immunity. New Phytol. 2018 Dec 15. doi: 10.1111/nph.15639.
Zhang et al. (2018). The MAP4 Kinase SIK1 Ensures Robust Extracellular ROS Burst and Antibacterial Immunity in Plants. Cell Host Microbe. 2018 Sep 12;24(3):379-391.e5. doi: 10.1016/j.chom.2018.08.007.
Lal et al. (2018). The Receptor-like Cytoplasmic Kinase BIK1 Localizes to the Nucleus and Regulates Defense Hormone Expression during Plant Innate Immunity. Cell Host Microbe. 2018 Apr 11;23(4):485-497.e5. doi: 10.1016/j.chom.2018.03.010. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Oxygenic photosynthesis poster by prof. Govindjee and Dr. Shevela
Z-scheme of photosynthetic electron transport by prof. Govindjee and Dr. Björn and Dr. Shevela - Reviews:
-
Fangling Yeh | 2025-01-02I have used this antibody for Western and coIP from the Arabidopsis seedling, which works well for both.Leon Zeng | 2021-01-06Works very well for our Western blot experiments.