1
Anti-LHCSR3
AS14 2766 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Bryopsis corticulans, Chlamydomonas reinhardtii, Nannochloropsis gaditana
- Product Info
-
Immunogen: KLH-conjugated synthetic peptide derived from LHCSR3 protein sequence from Chlamydomonas reinhardtii, UniProt: P0DO18
Host: Rabbit Clonality: Polyclonal Purity: Serum Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: ELISA (ELISA), Western blot (WB) Recommended dilution: 1 : 1000 (WB) Expected | apparent MW: 28 kDa
- Reactivity
-
Confirmed reactivity: Bryopsis corticulans, Chlamydomonas reinhardtii, Nannochloropsis gaditana Predicted reactivity: Phaeodactylum tricornutum
Species of your interest not listed? Contact usNot reactive in: Arabidopsis thaliana, Lobosphaera incisa, Marchantia polymorpha, Neochloris oleoabundans, Physcomitrella patens - Application Examples
-
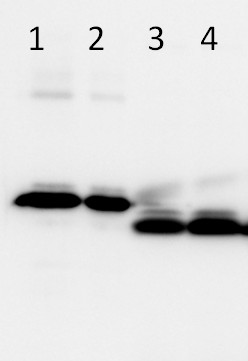
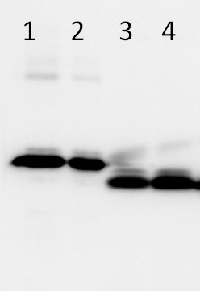
Follwoing samples: 0.1 µg of LhcSR3 IB + HisTag (1), 0.05 µg of LhcSR3 IB + HisTag (2), 5 µg of Chlamydomonas reinhardtii wild type (CC124) total protein extract of photoautotrophically grown cells in light intensity: 60 µE (3), 5 µg of Chlamydomonas reinhardtii wild type (CC124) total protein extract of photoautotrophically grown cells in high light intensity: 500 µE (4) were separated on 15% Tris-Glycine SDS PAGE and blotted overnight to PVDF using tank transfer. Blots were blocked with 5% BSA/milk for 1h at room temperature (RT) with agitation. Blot was incubated in the primary antibody at a dilution of 1: 1 000 for 1h at RT with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in PBS-T at RT with agitation. Blot was incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated, from Agrisera, AS09 602) diluted to 1:10 000 for 1h at RT with agitation. The blot was washed as above and developed for 5 min with ECL according to the manufacturer's instructions.
Courtesy Dr. Roberta Croce, Biophysics of Photosynthesis Dep. Physics and Astronomy Faculty of Sciences VU University Amsterdam, The Netherlands - Additional Information
-
Additional information (application): This antibody is also recognizing recombinant LHCSR1 overexpressed in E.coli as described in Perozeni et al. (2020). - Background
-
Background: LHCSR3 (Stress-related chlorophyll a/b binding protein 3) plays a role in an efficient enery dissipation process, called non-photochemical quenching (NPQ), in Chlamydomonas reinhardtii.
- Product Citations
-
Selected references: Blifernez-Klassen et al. (2026).The unicellular green microalga Botryosphaerella sudetica links plant-like light protection with an algal lifestyle. New Phytol. 2026 Feb 6. doi: 10.1111/nph.70990.
Liu et al. (2025). Hidden Diversity: Transcriptomic and Photosynthetic Variation Among Common" Wild Type" Chlamydomonas Strains. Plant J. 2025 Dec;124(5):e70615.doi: 10.1111/tpj.70615.
Damaraju et al. (2025). Investigating the role of PGR5 and PGRL1 in photosynthetic acclimation to salt stress in Chlamydomonas reinhardtii. Photosynth Res . 2025 Nov 5;163(6):58. doi: 10.1007/s11120-025-01179-w.
Arend et al. (2023) Widening the landscape of transcriptional regulation of green algal photoprotection. Nat Commun. 2023 May 10;14(1):2687. doi: 10.1038/s41467-023-38183-4. PMID: 37164999
Ruiz-Sola et al. (2023) Light-independent regulation of algal photoprotection by CO2 availability. Nat Commun. 2023 Apr 8;14(1):1977. doi: 10.1038/s41467-023-37800-6.
McQuilian et al. (2023). Proteomic characterization of a lutein-hyperaccumulating Chlamydomonas reinhardtii mutant reveals photoprotection-related factors as targets for increasing cellular carotenoid content. Biotechnol Biofuels Bioprod . 2023 Nov 4;16(1):166. doi: 10.1186/s13068-023-02421-0.
Bohmer et al. (2023) Chlamydomonas reinhardtii mutants deficient for Old Yellow Enzyme 3 exhibit increased photooxidative stress. Plant Direct. 2023;7(1):e480. Published 2023 Jan 15. doi:10.1002/pld3.480
Cazzaniga et al. (2022). Engineering astaxanthin accumulation reduces photoinhibition and increases biomass productivity under high light in Chlamydomonas reinhardtii. Biotechnol Biofuels Bioprod. 2022 Jul 11;15(1):77. doi: 10.1186/s13068-022-02173-3. PMID: 35820961; PMCID: PMC9277849.
Burlacot et al. (2022) Alternative photosynthesis pathways drive the algal CO2-concentrating mechanism. Nature 605, 366–371 (2022). https://doi.org/10.1038/s41586-022-04662-9
Cecchin et al (2021) LPA2 protein is involved in photosystem II assembly in Chlamydomonas reinhardtii. Plant J. 2021 Jul 4. doi: 10.1111/tpj.15405. Epub ahead of print. PMID: 34218480.
Roach et al. (2020). The non-photochemical quenching protein LHCSR3 prevents oxygen-dependent photoinhibition in Chlamydomonas reinhardtii. J Exp Bot. 2020 Jan 16. pii: eraa022. doi: 10.1093/jxb/eraa022.
Gabilly et al. (2019). Regulation of photoprotection gene expression in Chlamydomonas by a putative E3 ubiquitin ligase complex and a homolog of CONSTANS. Proc Natl Acad Sci U S A. 2019 Aug 12. pii: 201821689. doi: 10.1073/pnas.1821689116.
Tian et al. (2019). pH dependence, kinetics and light-harvesting regulation of nonphotochemical quenching in Chlamydomonas. Proc Natl Acad Sci U S A. 2019 Apr 23;116(17):8320-8325. doi: 10.1073/pnas.
Aihara et al. (2019). Algal photoprotection is regulated by the E3 ligase CUL4-DDB1DET1. Nat Plants. 2019 Jan;5(1):34-40. doi: 10.1038/s41477-018-0332-5.
Kong et al. (2018) Interorganelle Communication: Peroxisomal MALATE DEHYDROGENASE2 Connects Lipid Catabolism to Photosynthesis through Redox Coupling in Chlamydomonas. Plant Cell. 2018 Aug;30(8):1824-1847. doi: 10.1105/tpc.18.00361
Jokel et al. (2018). Hunting the main player enabling Chlamydomonas reinhardtii growth under fluctuating light. Plant J. 2018 Mar 25. doi: 10.1111/tpj.13897.
Kosuge et al.(2018). LHCSR1-dependent fluorescence quenching is mediated by excitation energy transfer from LHCII to photosystem I in Chlamydomonas reinhardtii. Proc Natl Acad Sci U S A. 2018 Apr 3;115(14):3722-3727. doi: 10.1073/pnas.1720574115.
Giovagnetti et al. (2018). A siphonous morphology affects light-harvesting modulation in the intertidal green macroalga Bryopsis corticulans (Ulvophyceae). Planta. 2018 Feb 19. doi: 10.1007/s00425-018-2854-5.
Chukhutsina et al. (2017). Photoprotection strategies of the alga Nannochloropsis gaditana. Biochim Biophys Acta. 2017 Jul;1858(7):544-552. doi: 10.1016/j.bbabio.2017.05.003.
Chaux et al. (2017). Flavodiiron Proteins Promote Fast and Transient O2 Photoreduction in Chlamydomonas. Plant Physiol. 2017 Jul;174(3):1825-1836. doi: 10.1104/pp.17.00421.
Wei et al. (2017). Light Intensity is Important for Hydrogen Production in NaHSO3-Treated Chlamydomonas reinhardtii. Plant Cell Physiol. 2017 Mar 1;58(3):451-457. doi: 10.1093/pcp/pcw216.
Garibay-Hernandez et al. (2016). Membrane proteomic insights into the physiology and taxonomy of an oleaginous green microalga. Plant Physiol. 2016 Nov 8. pii: pp.01240.2016. [Epub ahead of print]
Haraldsdottir (2016). Protection against UV rays and other desirable biological activity in Chlorella sp. and Phaeodactylum tricornutum. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Agrisera Educational Posters Collection - Reviews:
-
RANAY MOHAN YADAV | 2021-11-29working well in 1-1000 dilution
Sai Kiran Madireddi | 2018-10-04Species: C. reinhardtiiApplication: western blotDilution 1: 1000Result: successful.