1
Anti-LHY | Late elongated hypocotyl
AS13 2661 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Arabidopsis thaliana
- Product Info
-
Immunogen: KLH-conjugated synthetic peptide derived from Arabidopsis thaliana LHY protein sequence, UniProt: Q6R0H1-1, TAIR: AT1G01060
Host: Rabbit Clonality: Polyclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Western blot (WB) Recommended dilution: 1 : 1000 (WB) Expected | apparent MW: 70.4 | 88 kDa - Reactivity
-
Confirmed reactivity: Arabidopsis thaliana Predicted reactivity: Noccaea caerulescens
Species of your interest not listed? Contact usNot reactive in: Solanum tuberosum - Application Examples
-
Application example
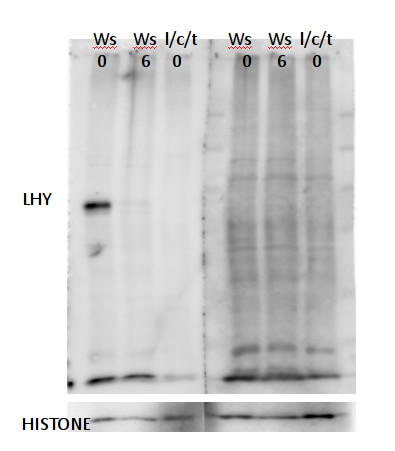
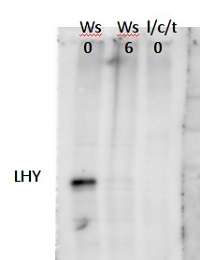
A clear band for LHY was detected at about 90kDa after electrophoresis using a 8% polyacrylamid gel cast in a Biorad gel device with 15 wells and 1mm thickness. Nuclear extract of Arabidopsis thaliana Ws (Wasilewska), zt0 zt6 and lhy/cca1/toc1 zt0 was analyzed and protein concentration was equalized according to Bradford quantification to ~10µg proteins per lane after resuspending and heating to 100°C for 10 min in SDS loading buffer. Proteins were well separated on the PAGE, the gels were equilibrated for 10 min in blotting buffer of 29g/L Glycine, 5,9g/L Tris base with 20% MEOH. Proteins were transferred in a wet western transfer device from Biorad for 12 h on a PVDF membrane. >From 50µg/50µl stock solution the antibody was diluted 1:500 in 1X TBS and incubated with or without 10µg/ml of peptide for 1h at RT followed by two hours incubation on separated parts of the same blot. Washing the unspecific bound antibody from the blot with TBS 0.1% tween for one hour changing solutions twice after the incubation. Secondary antibody was goat anti-rabbit HRP conjugated (AS09 602) used at 1:5000 in TBS with 5% milk for two hours shaking at RT. With TBS Tween unspecific bond antibody was washed of for one hour changing solution twice. For the last Wash TBS without tween was used for about 10 minutes. As a substrate for the HRP, high sensitivity chemiluminescent reagent was used and the emitted light detected with a FUJIFILM Las-3000 detecting pictures for 10 sec exposure with the increment setting. After 3 pictures taken a clear band was detectable.
HISTONE controls were done after stripping the blot, since LHY has a background band at about the same size as HISTONES.
Courtesy of Dr. Mark Ruhl, Umeå Plant Science Centre, Sweden - Additional Information
-
Additional information (application): Due to biology of LHY protein it accumulates short after dawn and not in the later day, therefore proper material has to be used to obtain a signal with this antibody.
Important note about protein extraction
Transcription factors are best isolated by freezing nucelar extract in liquid nitrogen after adding the extraction buffer, followed by thawing directly in 100°C heating block not in RT. When the samples were heated for 10 min, spinning of the extract for 5 min is needed to remove the cell debris. From the liquid extract measurement can be done and the rest, frozen in liquid Nitrogen, in aliquots. Frozen samples will not be thawed again to 100°C, but on RT and loaded as soon as they are liquid. Such precautions are necessary to take to be able to detect a transcription factor using antibodies.
This product can be sold containing ProClin if requested - Background
-
Background: LHY (Late elongated hypocotyl) is a transcription factor which is involved in the circadian clock. It binds to the promoter region of APRR1/TOC1 and TCP21/CHE to repress their transcription.
Alternative names: LATE ELONGATED HYPOCOTYL, LATE ELONGATED HYPOCOTYL 1, LHY, LHY1. - Protocols
-
- Reviews:
-
This product doesn't have any reviews.


