1
Agrisera BCIP/NBT Plus ALP Substrate (100 ml)
- Data sheet
-
Agrisera BCIP/NBT Plus ALP Substrate is designed to localize ALP labeled probes on immunohistochemical , in situ hybridization and blotting techniques. When the BCIP/NBT substrate solution is added to a blotting system containing ALP labeled probes the phosphate group of BCIP is hydrolyzed by ALP. This results in the formation of reducing components that reacts with NBT, converting it to insoluble purple NBT formazan visible on the blotting membrane.
Agrisera BCIP/NBT ALP Substrate contains BCIP (0.692 mMol L-1) and NBT (0.734 mMol L-1) in a 2-amino-2-methyl-1-propanol (AMP) buffer at a pH of 9,8 with added non-toxic stabilizers.
Features:
- Ready to use, single component BCIP/NBT reagent
- No to little background
- Contains non-toxic stabilizers
- Product Info
-
Quantity: 100 ml 1000 ml Storage: Store at 2 - 8 °C. - Application Examples
-
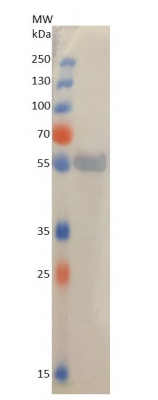
5 µg of total protein extracted freshly from Arabidopsis thaliana were separated on 15% SDS-PAGE and blotted 1 h to PVDF using semi-dry transfer. Blot was blocked with 5% milk in TBS-T 4°C/ON with agitation. Blot was incubated in the primary antibody at a dilution of 1: 5 000 for 1 h/RT with agitation in 5% milk in TBS-T with agitation. The antibody solution was decanted, and the blot was rinsed briefly, then washed 3 times for 5 min in TBS-T at RT with agitation. Blot was incubated in Agrisera matching secondary antibody (AS09 607 Goat anti-rabbit IgG, ALP conjugated) diluted to 1:1 000 in for 30 min/RT with agitation. The blot was washed 3 times for 10 min in TBS-T at RT with agitation and developed for 30 s with Agrisera BCIP/NBT ALP Substrate (AS19 BCIP-NBT-50).
- Additional Information
-
Additional information (application): Recommendation for use:
- Equilibrate reagent to room temperature before use.
- Following final binding reaction with an ALP labeled probe, wash the membrane in Tris-buffered saline or Tris/HCl containing 0,1% Tween 20.
- Do not use phosphate buffer for washing since this will inhibit ALP.
- Incubate the membrane in Agrisera BCIP/NBT ALP Substrate solution, protected from light, for 5-15 minutes.
- Make sure the membrane is completely covered in solution.
- Depending on enzyme activity, longer incubation may be necessary. If the concentration of ALP probe is too high, color might develop almost immediately, and the formazan deposit can flake off the membrane. High concentration of ALP probe can also result in formazan deposits forming a thin line around the band or dot, with no deposit in the center, This requires further dilution of the ALP probe.
- Wash the membrane with deionized water to stop the enzyme reaction.
- Dry the membrane and store at room temperature protected from light. Purple bands or dots will be visible at the sites of ALP activity, In case of excessive background staining try increasing the number of washes of the washing time since this indicates an incomplete removal of non-bound ALP from the membrane.
As soon as the desired band is detectable, briefly wash the membrane in generous amounts of deionized water. Transfer the membrane to fresh deionized water and incubate for 2 minutes with agitation. Change the water and incubate again for 5 minutes with agitation before placing the membrane on Whatman paper to dry. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts
Agrisera Educational Posters Collection - Reviews:
-
This product doesn't have any reviews.