1
Anti-ASyO5 | Mouse anti-human alpha-synuclein | oligomer-specific (clone number 2,4)
AS13 2718 | Clonality: monoclonal | Host: Mouse | Reactivity: Human
- Product Info
-
Sub class: IgG1 Immunogen: Synthetic peptide derived from human alpha-synuclein Gly111-Tyr125 Host: Mouse Clonality: Monoclonal Purity: Immunogen affinity purified serum in PBS pH 7.4. Format: Lyophilized Quantity: 50 µg Reconstitution: For reconstitution add 50 µl of sterile water Storage: For short time storage add sodium azide and store at +4°C. For long time storage store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Dot blot (Dot), ELISA (ELISA), Immunohistochemistry (IHC) Recommended dilution: 1-2 µg/ml (Dot), 2-4 µg/ml (ELISA capture), 10 µg/ml (IHC) Expected | apparent MW: 14 kDa
- Reactivity
-
Confirmed reactivity: Human, mouse - Application Examples
-
Application example
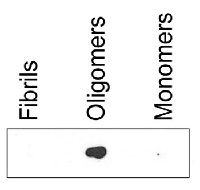
dot blot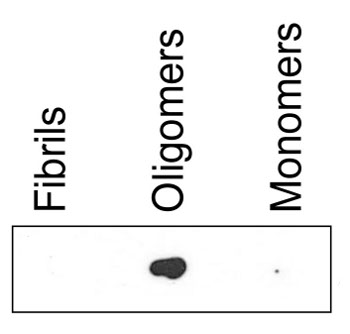
Dot blot reaction of the binding capacity of ASyO5 to fibrils, monomers and oligomers. Equal amounts of each sample were spotted on a nitrocellulose membrane and then dried. The membrane was blocked with 5% non-fat milk before incubated for 1 h with anti-ASyO5 (25nM) and then with secondary antibody, anti-mouse HRP-conjugated (1:1500). The membrane was washed with PBS containing 0.25% Tween-20 before detection using ECL prime (GE Healthcare).Preparation of α-synuclein oligomers and fibrils is described here.
Immunolocalization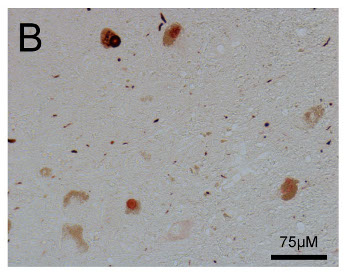
Tissue sections from the human PD midbrain, substantia nigra, were de-waxed and rehydrated in ethanol and then incubated with ASyO5 at RT for 1h. The immunoreactivity was detected with the anti-mouse Peroxidase Reagent Kit (ImmPRESS, Vector Laboratories, Inc.) and then developed using the ImmPACT AEC Peroxidase Substrate kit (Vector Laboratories, Inc.).
- Additional Information
-
Additional information: This antibody is specific to oligomers in ELISA as a capture antibody. For specific details, please check: Brännström et al. (2014). A Generic Method for Design of Oligomer-Specific Antibodies. PLoS ONE. DOI: 10.1371/journal.pone.0090857.
This antibody can detect the oligomer of A53T. - Background
-
Background: Alpha-synuclein is normally an unstructured soluble protein that can aggregate to form insoluble fibrils in pathological conditions characterized by Lewy bodies, such as Parkinson's disease, dementia with Lewy-bodies, and multiple system atrophy. In analogy to many other amyloid associated disorders, alpha-synuclein may also form oligomeric assemblies. These small and soluble forms have been suggested to exert a stronger tissue damaging effect as compared to the monomeric and fibrillar counterpart. Using a recently developed technique a monoclonal oligomer-specific antibody has been designed.
- Product Citations
-
Selected references: Kilpeläinen et al. (2025), Novel PREP ligand, HUP-46, ameliorates behavioral deficits in an alpha-synuclein based Parkinson's disease model. Exp Neurol. 2025 Dec 11:397:115593. doi: 10.1016/j.expneurol.2025.115593.
Kilpeläinen et al. (2025), Novel PREP ligand, HUP-46, ameliorates behavioral deficits in an alpha-synuclein based Parkinson's disease model. Exp Neurol. 2025 Dec 11:397:115593. doi: 10.1016/j.expneurol.2025.115593.
Kilpeläinen et al (2023) Nonpeptidic Oxazole-Based Prolyl Oligopeptidase Ligands with Disease-Modifying Effects on α-Synuclein Mouse Models of Parkinson's Disease.
Etelainen et al. (2022) Removal of proteinase K resistant alfaSyn species does not correlate with cell survival in a virus vector-based Parkinson's disease mouse model. Neuropharmacology. 2022;218:109213. doi:10.1016/j.neuropharm.2022.109213.
Ho et al. (2022). LRRK2 Inhibition Mitigates the Neuroinflammation Caused by TLR2-Specific alfa-Synuclein and Alleviates Neuroinflammation-Derived Dopaminergic Neuronal Loss. Cells. 2022 Mar 2;11(5):861. doi: 10.3390/cells11050861. PMID: 35269482; PMCID: PMC8909553.
Etelainen, Kilpelainen, Ignatius, et al. (2022) Removal of proteinase K resistant αSyn species does not correlate with cell survival in a virus vector-based Parkinson's disease mouse model. Neuropharmacology. 2022;218:109213. doi:10.1016/j.neuropharm.2022.109213.
Limegrover et al. (2021) Sigma-2 receptor antagonists rescue neuronal dysfunction induced by Parkinson's patient brain-derived a-synuclein. J Neurosci Res. 2021 Apr;99(4):1161-1176. doi: 10.1002/jnr.24782. Epub 2021 Jan 22. PMID: 33480104.
Kilpeläinen et al. (2019). Behavioural and dopaminergic changes in double mutated human A30P*A53T alpha-synuclein transgenic mouse model of Parkinson´s disease.Sci Rep. 2019 Nov 22;9(1):17382. doi: 10.1038/s41598-019-54034-z.
Wu et al. (2017). The critical role of Nramp1 in degrading a-synuclein oligomers in microglia under iron overload condition. Neurobiol Dis. 2017 Aug;104:61-72. doi: 10.1016/j.nbd.2017.05.001. (human, mouse, immunolocalization)
Svarcbahs et al. (2016). Inhibition of Prolyl Oligopeptidase Restores Spontaneous Motor Behavior in the a-Synuclein Virus Vector-Based Parkinson's Disease Mouse Model by Decreasing a-Synuclein Oligomeric Species in Mouse Brain. J Neurosci. 2016 Dec 7;36(49):12485-12497.
Brännström et al. (2014). A Generic Method for Design of Oligomer-Specific Antibodies. PLoS ONE. DOI: 10.1371/journal.pone.0090857. - Protocols
-
- Reviews:
-
This product doesn't have any reviews.

