1
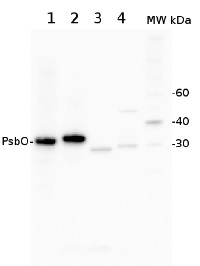
Discontinued PsbO | 33 kDa of the oxygen evolving complex (OEC) of PSII (anti-protein)
AS06 142-33 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A.thaliana, C. reinhardtii, C. vulgaris, C. sp. DT, E. crus-galli, F. arundinacea cv. Kord, H. pluvialis, H. spontaneum, H. vulgare, N. benthamiana, P. abies, P. banksiana, P. sativum, P. miliaceum, S. oleracea, S. tuberosum cultivar Taedong Valley, Synechococcus sp. PCC 7942 and PCC6803, T. salsuginea,
T.aestivum
Compartment marker of thylakoid lumen
Replaced by AS05 092
- Product Info
-
Immunogen: Native purified 33 kDa protein from Spinacia oleracea UniProt: P12359
Host: Rabbit Clonality: Polyclonal Purity: Total IgG. Protein G purified in PBS pH 7.4. Format: Lyophilized Quantity: 50 µl Reconstitution: For reconstitution add 50 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Immunoprecipitation (IP), Western blot (WB) Recommended dilution: 1 : 2000-1 : 5000 (WB) Expected | apparent MW: 35 | 33 kDa
- Reactivity
-
Confirmed reactivity: Arabidopsis thaliana, Chlamydomonas reinhardtii, Chlorella sp. DT, Chlorella vulgaris, Echinochloa crus-galli, Eucalyptus grandis x Eucalyptus camaldulensis, Festuca arundinacea cv. Kord, Haematococcus pluvialis, Hordeum spontaneum, Hordeum vulgare, Nicotiana benthamiana, Oryza sativa, Panicum miliaceum, Picea abies, Pinus banksiana, Pisum sativum, Spinacia oleracea, Solanum tuberosum cultivar Taedong Valley, Synechococcus sp. PCC7002, sp. PCC 7942 and sp. PCC6803, Thellungiella salsuginea, Triticum aestivum, Zea mays
Predicted reactivity: Galdieria sulphuraria, Glycine max, Nicotiana tabacum, Solanum lycopersicum, Populus trichocarpa, Zosteria marina
Species of your interest not listed? Contact usNot reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
Application example
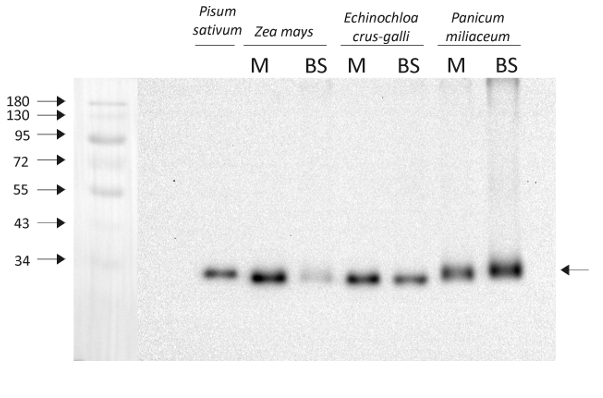
1 µg of chlorophyll from Pisum sativum thylakoids and from mesophyll (M) and bundle sheath (BS) thylakoids of Zea mays, Echinochloa crus-galli, Panicum miliaceum extracted with 0.4 M sorbitol, 50 mM Hepes NaOH, pH 7.8, 10 mM NaCl, 5 mM MgCl2 and 2 mM EDTA were loaded to lanes. Samples were denatured with Laemmli buffer at 75 0C for 5 min and were separated on 12% SDS-PAGE and blotted 30 min to PVDF using wet transfer. Blot was blocked with 5% milk for 2h at room temperature (RT) with agitation. Blot was incubated in the primary antibody AS06 142-33 (Lot 1902) at a dilution of 1: 2000 overnight at 40C with agitation in 1% milk in TBS-T. The antibody solution was decanted and the blot was washed 4 times for 5 min in TBS-T at RT with agitation. Blot was incubated in secondary antibody (anti-rabbit IgG HRP conjugated, from Agrisera, AS09 602, Lot 1902, as recommended) diluted to 1:20 000 in 1 % milk in TBS-T for 1h at RT with agitation. The blot was washed 5 times for 5 min in TBS-T and 2 times for 5 min in TBS, and developed for 1 min with 1.25 mM luminol, 0.198 mM coumaric acid and 0.009% H2O2 in 0.1 M Tris- HCl, pH 8.5. Exposure time in ChemiDoc System was 100 seconds
Courtesy of Dr. Wioleta Wasilewska-Dębowska, Warsaw University, Poland
2 µg of total protein from Arabidopsis thaliana leaf (1), Hordeum vulgare leaf (2), Chlamydomonas reinhardtii total cell (3), Synechococcus sp. 7942 total cell (4), all extracted with PEB (AS08 300) were separated on 4-12% NuPage (Invitrogen) LDS-PAGE and blotted 1h to PVDF. Blots were blocked immediately following transfer in 2% ECL Advance blocking reagent in 20 mM Tris, 137 mM sodium chloride pH 7.6 with 0.1% (v/v) Tween-20 (TBS-T) for 1h at room temperature with agitation. Blots were incubated in the primary antibody at a dilution of 1: 10 000 for 1h at room temperature with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in TBS-T at room temperature with agitation. Blots were incubated in secondary antibody (anti-rabbit IgG horse radish peroxidase conjugated) diluted to 1:50 000 in 2% blocking solution for 1h at room temperature with agitation. The blots were washed as above and developed for 5 min with chemiluminescent detection reagent according the manufacturers instructions. Images of the blots were obtained using a CCD imager (FluorSMax, Bio-Rad) and Quantity One software (Bio-Rad).
Note: Detection level in algal species can be improved by adjustment of western blot protocol. Please inquire.Application examples: 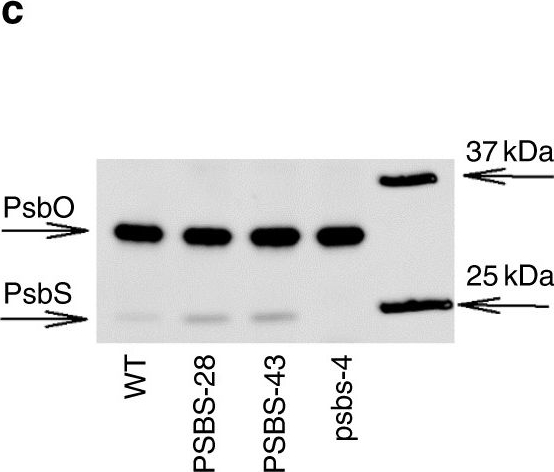
Reactant: Nicotiana tabacum (Common tobacco)
Application: Western Blotting
Pudmed ID: 29511193
Journal: Nat Commun
Figure Number: 1C
Published Date: 2018-03-06
First Author: G?owacka, K., Kromdijk, J., et al.
Impact Factor: 13.783
Open PublicationPhotosynthesis and water-use efficiency in Nicotiana tabacum plants with modified PsbS levels. aPsbS mRNA levels normalized to actin and tubulin sampled from fully expanded leaves of psbs-4, PSBS-28, PSBS-43 and wild-type (WT) N. tabacum plants. b PsbS protein levels normalized to the large subunit of oxygen-evolving complex of photosystem II (PsbO), determined from densitometry on immunoblots. c Representative immunoblot for PsbS and PsbO. d Net CO2 fixation rate (An), e NPQ, f quinone A (QA) redox state, and g stomatal conductance (gs) as a function of incident light intensity in fully expanded leaves. h Linear correlation between QA redox state and gs. Broken lines indicate measurements at highest light intensity. i Linear correlation between PsbS protein levels and intrinsic water-use efficiency (An/gs) at light intensity above 600??mol?m?2?s?1. Asterisks/lines show significant differences from WT (black for silencing, red for overexpressing lines; Dunnett’s two-way test; ??=?0.05). Error bars indicate s.e.m. (n?=?from 6 to 10 biological replicates)
- Additional Information
-
Additional information: Total IgG fraction has been purified by 40% ammonium sulpgate precipitation followed by DEAE cellulose chromatography
This product can be sold containing ProClin if requestedAdditional information (application): This antibody can be used as a loading control for Chlamydomonas reinhardtii while it not so suitable for higher plants as accumulation of these proteins might drop to 12.5-25 % of the WT level in mutants defective for PSII core (Schult et al. 2007).
- Background
-
Background: PSII reaction centre components are generating the redox potential required to drive highly oxidizing water splitting reaction. Four Mn atoms are present on a lumenal surface and form the catalyctic site of the water-splitting reaction which is in close association with the 33 kDa (PsbO), 23 kDa (PsbP) and 17 kDa (PsbQ) extrinistic subunits of oxygen evolving complex OEC. A 33-kDa extrinsic protein is also termed the Mn-stabilizing protein (MSP), however recent evidences shown that it is C-terminal domain of PsbA (D1) protein which is involved in in the assembly and stabilization of the OEC.
- Product Citations
-
Selected references: Turc et al. (2024). Non-photochemical quenching upregulation improves water use efficiency and reduces whole plant level water consumption under drought. J Exp Bot. 2024 Mar 12:erae113. doi: 10.1093/jxb/erae113.
Nagy et al. (2023). Photoautotrophic and sustained H2 production by the pgr5 mutant of Chlamydomonas reinhardtii in simulated daily light conditions. International Journal of Hydrogen Energy Volume 53, 31 January 2024, Pages 760-769.
Cecchin et al (2021) LPA2 protein is involved in photosystem II assembly in Chlamydomonas reinhardtii. Plant J. 2021 Jul 4. doi: 10.1111/tpj.15405. Epub ahead of print. PMID: 34218480.
Loudya et al. (2021) Cellular and transcriptomic analyses reveal two-staged chloroplast biogenesis underpinning photosynthesis build-up in the wheat leaf. Genome Biol. 2021 May 11;22(1):151. doi: 10.1186/s13059-021-02366-3. PMID: 33975629; PMCID: PMC8111775.
Terentyev (2020: The Main Structural and Functional Characteristics of Photosystem-II-Enriched Membranes Isolated From Wild Type and cia3 Mutant Chlamydomonas reinhardtii. Life (Basel). 2020 May 14;10(5):E63. doi: 10.3390/life10050063..
Tang el al. (2020). OsNSUN2-Mediated 5-Methylcytosine mRNA Modification Enhances Rice Adaptation to High Temperature. Dev Cell. 2020 May 4;53(3):272-286.e7. doi: 10.1016/j.devcel.2020.03.009.
Smythers et al. (2019). Characterizing the effect of Poast on Chlorella vulgaris, a non-target organism. Chemosphere Volume 219, March 2019, Pages 704-712. - Protocols
-
Agrisera Western Blot protocol and video tutorials
Protocols to work with plant and algal protein extracts - Reviews:
-
Soujanya Kuntam | 2021-11-09We used this antibody to detect PsbO in complete protein extracts of C. reinhardtii using western blotting. It worked well without much optimization needed.Amanda Smythers | 2020-12-03I used this antibody to help confirm psbO degradation in PSII-enriched membranes from spinach. It was easy to use and worked perfectly without optimization and helped lead to our publication: doi: 10.1016/j.chemosphere.2018.12.050Katya Georgieva | 2019-06-24I used the antibody to check its expression during desiccation of Haberlea rhodopensis. PsbO is working perfect (1:2000 dilution) on isolated thylakoids and total leaf proteins. Clear, single band is obtained with ECL development.Qing-Bo | 2018-11-23I used the antibody to detect the accumulation of the corresponding
in Arabidopsis thaliana mutant with a dilution 1: 1000. The antibody works very well.Jun Liu | 2014-01-146 µg of total protein from Arabidopsis was separated on 12 % SDS-page Gel , blotted 1h to NC membrane. Blots were incubated in the primary antibody at a dilution of 1: 20 000 overnight, A clear and specific band above 30 kDa has always been detected by ECL .Jerzy Kruk | 2013-11-21Analysis perfomed on Arabidopsis total leaf extract, 10% SDS gel, 3 ug chlorophyll per lane, AB dilution 1:2000, the product observed at ca. 33 kDaMayank Anand | 2010-06-16I am satisfied with results I got, usinf Agrisera antibody. I used a 1:15000 dilution for this antibody. I am checking its expression at different stages of potato tuberization, so I took proteins from the stolons, initial tuber, maturing tubers and mature tubers.Irma Roberts | 2009-05-19I used at a dilution of 1:20000 plus 1:150000 for the secondary antibody against PsbO from spinach, developing the membrane with ECL advance solution from GE.
Related products
AS06 142-23 | Clonality: Polyclonal | Host: Rabbit | Reactivity: A. thaliana, Ch. reinhardtii, P. banksiana. , S.oleracea, T. aestivum


