1
Anti-HSP70/HSC70 | Heat shock protein 70/Heat shock cognate protein 70 (serum)
AS05 083 | Clonality: Polyclonal | Host: Rabbit | Reactivity: [global abtibody] for HSP70 and HSC70 in fish, mammals and fungi
Benefits of using this antibody
- Product Info
-
Immunogen: KLH-conjugated synthetic peptide conserved across all known sequences of HSP70 P08107 and HSC70 proteins P11142
Host: Rabbit Clonality: Polyclonal Purity: Total IgG. Protein G purified in PBS pH 7.4. Format: Lyophilized Quantity: 100 µl Reconstitution: For reconstitution add 100 µl of sterile water Storage: Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. Tested applications: Immunoprecipitation (IP), Western blot (WB) Recommended dilution: 1 : 1000 (IP), 1 : 1000-1: 5000 (WB) Expected | apparent MW: 70 kDa
- Reactivity
-
Confirmed reactivity: Fish, mammals, fungi: Antrodia infirma, A. sinuosa, A. xantha, Catostomus commersonii, Gloeophyllum protractum, Gloeophyllum sepiarium, G. carbonarium, Junghunia luteoalba, Oligoporus sericiomollis, Phlebia cornea Predicted reactivity: Bovine, Drosophila melanogaster, Hen, Mouse, Rat Not reactive in: No confirmed exceptions from predicted reactivity are currently known - Application Examples
-
Application example
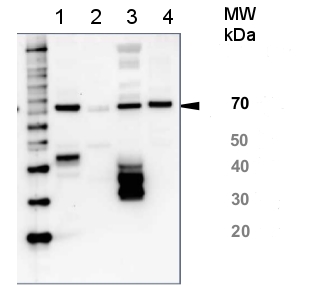
10 µg of total protein from (1) killi fish muscle, (2) bovine muscle, (3) chicken muscle, (4) rat liver, extracted with Protein Extration Buffer, PEB (AS08 300) and separated on 4-12% NuPage (Invitrogen) LDS-PAGE and blotted 1h to PVDF. Blots were blocked in 5 % non-fat milk for 1h at room temperature with agitation. Blots were incubated in the primary antibody at a dilution of 1: 5000 (in blocking reagent) for 1h at room temperature with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in TBS-T at room temperature with agitation. Blots were incubated in secondary antibody (Agrisera anti-rabbit IgG horse radish peroxidase conjugated, AS09 602) diluted to 1:25 000 in blocking reagent for 1h at room temperature with agitation. The blots were washed as above and developed for 5 min with chemiluminescent detection reagent, according to the manufacturers instructions. Images of the blots were obtained using a CCD imager (FluorSMax, Bio-Rad) and Quantity One software (Bio-Rad). Exposure time was 5 min.
- Additional Information
-
Additional information: For detection of plant and algal cytoplasmic hsp70 we recommend following product: AS08 371.
Additional information (application): This antibody is recognizing both, the inducible and the constitutive Hsp70 - Background
-
Background: Heat shock protein 70 (Hsp70) is the major stress-inducible protein in vertebrates and is highly conserved throughout evolution. It plays a role as a molecular chaperone and is important for allowing cells to cope with acute stress or insult, especially those affecting the protein machinery. Heat shock cognate protein 70 (HSC70) is a highly conserved protein and a member of the family of molecular chaperones. Alternative names: HSP70.1, HSP70-1/HSP70-2, Heat shock 70 kDa protein 8
- Product Citations
-
Selected references: MacLellan et al. (2015). Chaperone roles for TMAO and HSP70 during hyposmotic stress in the spiny dogfish shark (Squalus acanthias). J Comp Physiol B. 2015 Jun 7.
Bessemer et al. (2014). Cardiorespiratory toxicity of environmentally relevant zinc oxide nanoparticles in the freshwater fish Catostomus commersonii. Nanotoxicology. 2014 Nov 27:1-10.
Gorovits et al. (2013). Recruitment of the host plant heat shock protein 70 by tomato yellow leaf curl virus coat protein is required for virus infection. PLoS Once, July 23;8(7).
- Protocols
- Antibody protocols
- Reviews:
-
This product doesn't have any reviews.
Related products

AS05 083A | Clonality: Polyclonal | Host: Rabbit | Reactivity: [global abtibody] for HSP70 and HSC70 in fish, mammals and fungi
Benefits of using this antibody


